

Name: Glycerin
CAS No: Propane-1,2,3-triol [56-81-5]
BP: Glycerol JP: Concentrated Glycerin PhEur: Glycerol USP: Glycerin
Croderol; E422; glicerol; glycerine; glycerolum; Glycon G-100; Kemstrene; Optim; Pricerine; 1,2,3-propanetriol; trihydroxypropane glycerol.
Propane-1,2,3-triol [56-81-5]
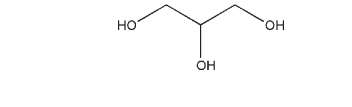
C3H8O3 , 92.09
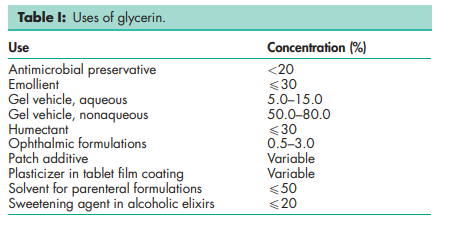
Glycerin is used in a wide variety of pharmaceutical formulations including oral, otic, ophthalmic, topical, and parenteral preparations; see Table I. In topical pharmaceutical formulations and cosmetics, glycerin is used primarily for its humectant and emollient properties. Glycerin is used as a solvent or cosolvent in creams and emulsions.(1–3) Glycerin is additionally used in aqueous and nonaqueous gels and also as an additive in patch applications.(4–6) In parenteral formulations, glycerin is used mainly as a solvent and cosolvent.(7–10) In oral solutions, glycerin is used as a solvent,(10) sweetening agent, antimicrobial preservative, and viscosity-increasing agent. It is also used as a plasticizer and in film coatings.(11–14) Glycerin is used as a plasticizer of gelatin in the production of soft-gelatin capsules and gelatin suppositories. Glycerin is employed as a therapeutic agent in a variety of clinical applications,(15) and is also used as a food additive.
Glycerin is a clear, colorless, odorless, viscous, hygroscopic liquid; it has a sweet taste, approximately 0.6 times as sweet as sucrose.
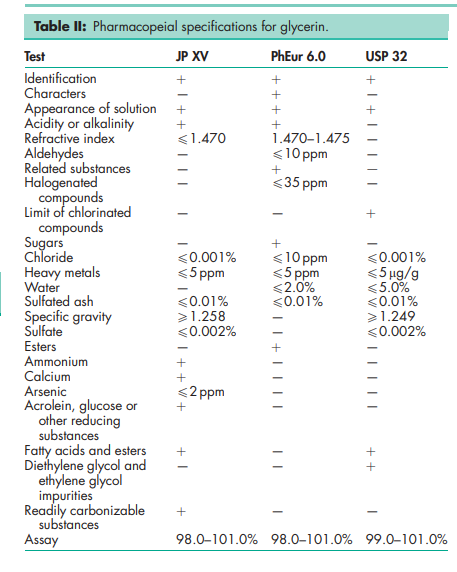
See Table II.
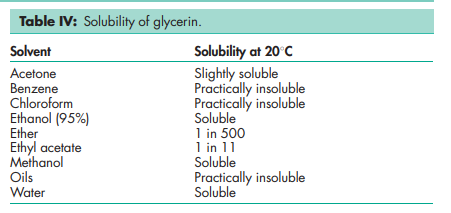
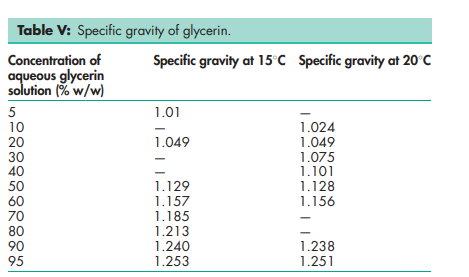
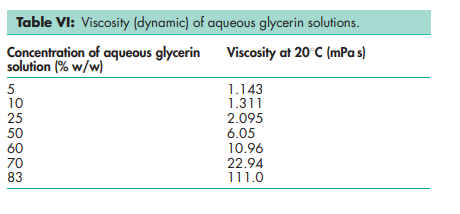
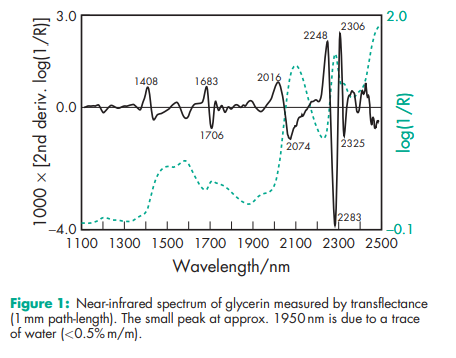
Boiling point 2908C (with decomposition) Density 1.2656 g/cm3 at 158C; 1.2636 g/cm3 at 208C; 1.2620 g/cm3 at 258C. Flash point 1768C (open cup) Freezing point see Table III. Hygroscopicity Hygroscopic. Melting point 17.88C NIR spectra see Figure 1. Osmolarity A 2.6% v/v aqueous solution is isoosmotic with serum. Refractive index n D 15 = 1.4758; n D 20 = 1.4746; n D 25 = 1.4730. Solubility see Table IV. Specific gravity see Table V. Surface tension 63.4 mN/m (63.4 dynes/cm) at 208C. Vapor density (relative) 3.17 (air = 1) Viscosity (dynamic) see Table VI

Glycerin is hygroscopic. Pure glycerin is not prone to oxidation by the atmosphere under ordinary storage conditions, but it decomposes on heating with the evolution of toxic acrolein. Mixtures of glycerin with water, ethanol (95%), and propylene glycol are chemically stable. Glycerin may crystallize if stored at low temperatures; the crystals do not melt until warmed to 208C. Glycerin should be stored in an airtight container, in a cool, dry place
Glycerin may explode if mixed with strong oxidizing agents such as chromium trioxide, potassium chlorate, or potassium permanganate. In dilute solution, the reaction proceeds at a slower rate with several oxidation products being formed. Black discoloration of glycerin occurs in the presence of light, or on contact with zinc oxide or basic bismuth nitrate. An iron contaminant in glycerin is responsible for the darkening in color of mixtures containing phenols, salicylates, and tannin. Glycerin forms a boric acid complex, glyceroboric acid, that is a stronger acid than boric acid.
Glycerin is mainly obtained from oils and fats as a by-product in the manufacture of soaps and fatty acids. It may also be obtained from natural sources by fermentation of, for example, sugar beet molasses in the presence of large quantities of sodium sulfite. Synthetically, glycerin may be prepared by the chlorination and saponification of propylene.
Glycerin occurs naturally in animal and vegetable fats and oils that are consumed as part of a normal diet. Glycerin is readily absorbed from the intestine and is either metabolized to carbon dioxide and glycogen or used in the synthesis of body fats. Glycerin is used in a wide variety of pharmaceutical formulations including oral, ophthalmic, parenteral, and topical preparations. Adverse effects are mainly due to the dehydrating properties of glycerin.(15) Oral doses are demulcent and mildly laxative in action. Large doses may produce headache, thirst, nausea, and hyperglycemia. The therapeutic parenteral administration of very large glycerin doses, 70–80 g over 30–60 minutes in adults to reduce cranial pressure, may induce hemolysis, hemoglobinuria, and renal failure.(16) Slower administration has no deleterious effects.(17) Glycerin may also be used orally in doses of 1.0–1.5 g/kg bodyweight to reduce intraocular pressure. When used as an excipient or food additive, glycerin is not usually associated with any adverse effects and is generally regarded as a nontoxic and nonirritant material. LD50 (guinea pig, oral): 7.75 g/kg(18) LD50 (mouse, IP): 8.70 g/kg LD50 (mouse, IV): 4.25 g/kg LD50 (mouse, oral): 4.1 g/kg LD50 (mouse, SC): 0.09 g/kg LD50 (rabbit, IV): 0.05 g/kg LD50 (rabbit, oral): 27 g/kg(19) LD50 (rat, IP): 4.42 g/kg LD50 (rat, oral): 5.57 g/kg(19) LD50 (rat, oral): 12.6 g/kg LD50 (rat, SC): 0.1 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended. In the UK, the recommended long-term (8-hour TWA) workplace exposure limit for glycerin mist is 10 mg/m3 . (20) Glycerin is combustible and may react explosively with strong oxidizing agents.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (dental pastes; buccal preparations; inhalations; injections; nasal and ophthalmic preparations; oral capsules, solutions, suspensions and tablets; otic, rectal, topical, transdermal, and vaginal preparations). Included in nonparenteral and parenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients
—
Glycerin is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. Some pharmacopeias also contain specifications for diluted glycerin solutions. The JP XV contains a monograph for ‘glycerin’ that contains 84–87% of propane-1,2,3-triol (C3H8O3). The PhEur 6.0 contains a monograph for ‘glycerol 85 per cent’ that contains 83.5–88.5% of propane-1,2,3-triol (C3H8O3). A specification for glycerin is contained in the Food Chemicals Codex (FCC).(21) The EINECS number for glycerin is 200-289-5.