

Name: Glyceryl Behenate
CAS No: Docosanoic acid, monoester with glycerin [30233-64-8] (glycerylbehenate) , diester with glycerin [94201-62-4] (glyceryldibehenate), triester with glycerin[18641-57-1] (glyceryltribehenate)
BP: Glycerol Dibehenate PhEur: Glycerol Dibehenate USP-NF: Glyceryl Behenate
Compritol 888 ATO; 2,3-dihydroxypropyl docosanoate; docosanoic acid, 2,3-dihydroxypropyl ester; E471; glycerol behenate; glyceroli dibehenas; glyceryl monobehenate. Note that tribehenin is used as a synonym for glyceryl tribehenate
Docosanoic acid, monoester with glycerin [30233-64-8] (glycerylbehenate) , diester with glycerin [94201-62-4] (glyceryldibehenate), triester with glycerin[18641-57-1] (glyceryltribehenate)
Glyceryl dibehenate is a mixture of glycerol esters. The PhEur 6.0 describes glyceryl dibehenate as a mixture of diacylglycerols, mainly dibehenoylglycerol, together with variable quantities of mono- and triacylglycerols (see Section 9). The USP32–NF27 describes glyceryl behenate as a mixture of glycerides of fatty acids, mainly behenic acid. It specifies that the content of 1-monoglycerides should be 12.0–18.0%
Glyceryl behenate is used in cosmetics, foods, and oral pharmaceutical formulations; see Table I. In pharmaceutical formulations, glyceryl behenate is mainly used as a lubricant in the preparation of oral tablets and capsules.(1–3) It has good binding properties, it does not affect tablet hardness and is unaffected by mixing or production parameters. Glyceryl behenate has been investigated for the encapsulation of various drugs such as retinoids.(4) It has also been investigated for use in the preparation of sustained-release tablets;(5–10) as a matrix-forming agent for the controlled release of water-soluble drugs;(10) and it can also be used as a hot-melt coating agent sprayed onto a powder or drug-loaded sugar beads and granules.(11,12) It may also be incorporated via extrusion/spheronization into pellets, which can be further compressed into tablets. Glyceryl behenate is used in oral enteric-coated pellets, powders and suspensions. It is also used in controlled, extended-release and orally disintegrating tablets. For oral preparations, glyceryl behenate forms a lipidic matrix for sustained-release formulations. It has been used along with acid-soluble or swellable polymers to mask the bitter or unpleasant taste of the medicament with improved palatability.(13) Glyceryl behenate has been used for the preparation of ophthalmic inserts.(14,15) In cosmetics, glyceryl behenate is used as a skin conditioning agent, emollient and viscosity-increasing agent in emulsions. It also improves the heat stability of emulsions and is a gelifying agent for various oils. For topical formulations, it is used as a thickening agent for oily phases. It is also used as a surfactant or emulsifying agent

Glyceryl behenate occurs as a fine white-yellow powder, as a hard waxy mass or pellet, or as white or almost white unctuous flakes. It has a faint odor.
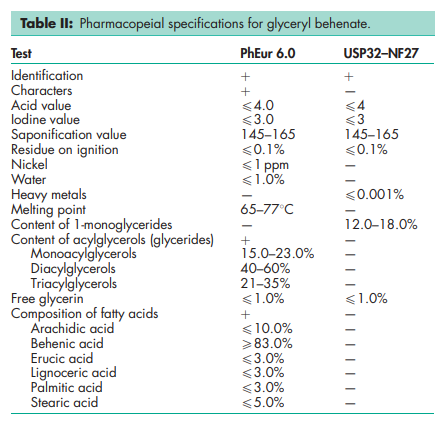
See Table II
HLB value 2 Melting point 65–778C Solubility Soluble, when heated, in chloroform and dichloromethane and in many organic solvents; slightly soluble in hot ethanol (96%); practically insoluble in cold ethanol (95%), hexane, mineral oil, and water.
Glyceryl behenate should be stored in a tightly closed container, at a temperature less than 358C.
—
Glyceryl behenate is prepared by the esterification of glycerin by behenic acid (C22 fatty acid) without the use of catalysts. In the case of Compritol 888 ATO (Gattefosse´), raw materials used are of vegetable origin, and the esterified material is atomized by spraycooling.
Glyceryl behenate is used in cosmetics, foods and oral pharmaceutical formulations, and is generally regarded as a relatively nonirritant and nontoxic material. The US Cosmetic Ingredients Review Expert Panel evaluated glyceryl behenate and concluded that it is safe for use in cosmetic formulations in present practices of use and concentration. LD50 (mouse, oral): 5 g/kg(16)
Observe normal precautions appropriate to the circumstances and quantities of material handled. Glyceryl behenate emits acrid smoke and irritating fumes when heated to decomposition.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (oral capsules, tablets, and suspensions). Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Glyceryl palmitostearate.
Glyceryl behenate is an amphiphilic material with a high melting point and, therefore, has been investigated in the preparation of aqueous colloidal dispersions such as solid lipid microparticles (SLM), nanoparticles (SLN) and nanostructured lipid carriers (NLC) for the entrapment of lipophilic drugs.(17) For example, SLM of ibuprofen have been prepared with a high drug-loading capacity.(18–20) Glyceryl behenate SLM for the sunscreen agent octyl-dimethylaminobenzoate (ODAB), showed enhanced sunscreen photostability, and SLM loaded with the polar adenosine A1 receptor agonist N6 -cyclopentyladenosine (CPA) have shown improved drug stability.(21,22) SLM have been prepared using glyceryl behenate as carriers for pulmonary delivery.(23) SLN prepared from glyceryl behenate have been investigated for oral and mucosal delivery with enhanced bioavailability;(24,25) for topical and transdermal delivery of vitamin A,(26) retinoic acid,(27) ketoconazole,(28) ketorolac,(29) and for parenteral drug administration of drugs such as tetracaine, etomidate and prednisolone.(30–33) NLC have been prepared by replacing part of solid lipid by oil for improved encapsulation of drugs.(34) The EINECS numbers are: 250-097-0 for glyceryl behenate; 303-650-6 for glyceryl dibehenate; 242-471-7 for glyceryl tribehenate. The PubChem Compound ID (CID) for glyceryl behenate includes 62726 and 121658.