

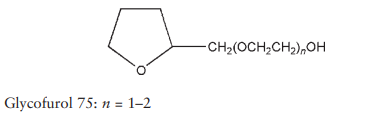
Name: Glycofurol
CAS No: a-[(Tetrahydro-2-furanyl)methyl]-o-hydroxy-poly(oxy-1,2-ethanediyl) [31692-85-0]
None adopted
Glycofurol 75; tetraglycol; a-(tetrahydrofuranyl)-o-hydroxy-poly(- oxyethylene); tetrahydrofurfuryl alcohol polyethylene glycol ether; THFP. Note: tetraglycol is also used as a synonym for tetrahydrofurfuryl alcohol.
a-[(Tetrahydro-2-furanyl)methyl]-o-hydroxy-poly(oxy-1,2-ethanediyl) [31692-85-0]
C9H18O4 (average), 190.24 (average)
Glycofurol is used as a solvent in parenteral products for intravenous or intramuscular injection in concentrations up to 50% v/v.(1–5) It has also been investigated, mainly in animal studies, for use as a penetration enhancer and solvent in topical(6) and intranasal formulations.(7–11) Glycofurol has also been used at 20% v/v concentration in a rectal formulation.(12
Glycofurol is a clear, colorless, almost odorless liquid, with a bitter taste; it produces a warm sensation on the tongue.
—
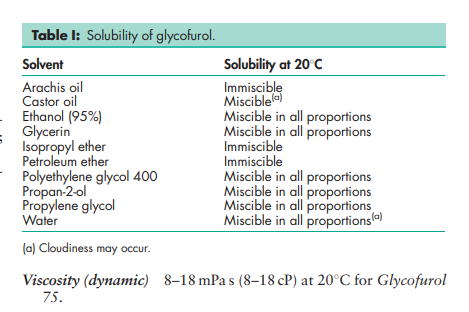
Boiling point 80–1008C for Glycofurol 75 Density 1.070–1.090 g/cm3 at 208C Hydroxyl value 300–400 Moisture content 0.2–5% at ambient temperature and 30% relative humidity. Refractive index n D 40 = 1.4545 Solubility see Table I.
Stable if stored under nitrogen in a well-closed container protected from light, in a cool, dry place.
Incompatible with oxidizing agents.
Glycofurol is prepared by the reaction of tetrahydrofurfuryl alcohol with ethylene oxide (followed by a special purification process in the case of Glycofurol 75).
Glycofurol is mainly used as a solvent in parenteral pharmaceutical formulations and is generally regarded as a relatively nontoxic and nonirritant material at the levels used as a pharmaceutical excipient. Glycofurol can be irritant when used undiluted; its tolerability is approximately the same as propylene glycol.(1,2) Glycofurol may have an effect on liver function and may have a low potential for interaction with hepatoxins or those materials undergong extensive hepatic metabolism.(4) LD50 (mouse, IV): 3.5 mL/kg(1,2)
Observe normal precautions appropriate to the circumstances and quantity of material handled.
Included in parenteral medicines licensed in Europe.
—
Grades other than Glycofurol 75 may contain significant amounts of tetrahydrofurfuryl alcohol and other impurities. Glycofurol 75 meets an analytical specification which includes a requirement that the fraction in which n = 1 or 2 amounts to a minimum of 95%; see Section 5.