

Name: Xanthan Gum
CAS No: Xanthan gum [11138-66-2]
BP: Xanthan Gum PhEur: Xanthan Gum USP-NF: Xanthan Gum
Corn sugar gum; E415; Grindsted; Keldent; Keltrol; polysaccharide B-1459; Rhodicare S; Rhodigel; Vanzan NF; xanthani gummi; Xantural.
Xanthan gum [11138-66-2]
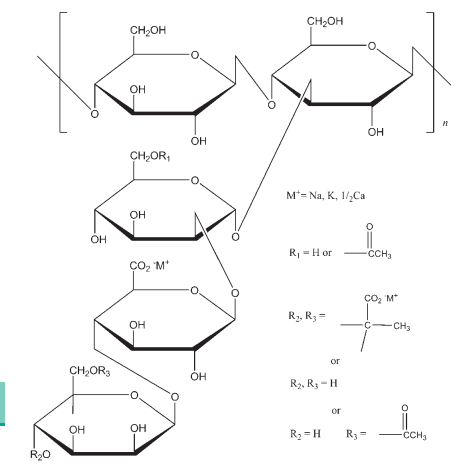
(C35H49O29)n approximately 1 106 The USP32–NF27 describes xanthan gum as a high molecular weight polysaccharide gum. It contains D-glucose and D-mannose as the dominant hexose units, along with D-glucuronic acid, and is prepared as the sodium, potassium, or calcium salt. Each xanthan gum repeat unit contains five sugar residues: two glucose, two mannose, and one glucuronic acid. The polymer backbone consists of four b-D-glucose units linked at the 1 and 4 positions, and is therefore identical in structure to cellulose. Trisaccharide side chains on alternating anhydroglucose units distinguish xanthan from cellulose. Each side chain comprises a glucuronic acid residue between two mannose units. At most of the terminal mannose units is a pyruvate moiety; the mannose nearest the main chain carries a single group at C-6. The resulting stiff polymer chain may exist in solution as a single, double, or triple helix that interacts with other xanthan gum molecules to form complex, loosely bound networks.(1,
Xanthan gum is widely used in oral and topical pharmaceutical formulations, cosmetics, and foods as a suspending and stabilizing agent.(3–5) It is also used as a thickening and emulsifying agent. It is nontoxic, compatible with most other pharmaceutical ingredients, and has good stability and viscosity properties over a wide pH and temperature range; see Section 11. Xanthan gum gels show pseudoplastic behavior, the shear thinning being directly proportional to the shear rate. The viscosity returns to normal immediately on release of shear stress. Xanthan gum has been used as a suspending agent for conventional,(6) dry(7) and sustained-release(8) suspensions. When xanthan gum is mixed with certain inorganic suspending agents, such as magnesium aluminum silicate, or organic gums, synergistic rheological effects occur.(9) In general, mixtures of xanthan gum and magnesium aluminum silicate in ratios between 1 : 2 and 1 : 9 produce the optimum properties. Similarly, optimum synergistic effects are obtained with xanthan gum : guar gum ratios between 3 : 7 and 1 : 9. Although primarily used as a suspending agent, xanthan gum has also been used to prepare sustained-release matrix tablets.(10–13) Controlled-release tablets of diltiazem hydrochloride prepared using xanthan gum have been reported to sustain the drug release in a predictable manner, and the drug release profiles of these tablets were not affected by pH and agitation rate.(14) Xanthan gum has also been used to produce directly compressed matrices that display a high degree of swelling due to water uptake, and a small amount of erosion due to polymer relaxation.(15) It has also been used in combination with chitosan,(16,17) guar gum,(18,19) galactomannan,(20) and sodium alginate(21) to prepare sustained-release matrix tablets. Xanthan gum has been used as a binder,(22) and in combination with Konjac glucomannan(23,24) is used as an excipient for controlled colonic drug delivery. Xanthan gum with boswellia (3 : 1)(25) and guar gum (10 : 20)(26) have shown the best release profiles for the colon-specific compression coated systems of 5- fluorouracil for the treatment of colorectal cancer. Xanthan gum has also been used with guar gum for the development of a floating drug delivery system.(27) It has also has derivatized to sodium carboxymethyl xanthan gum and crosslinked with aluminum ions to prepare microparticles, as a carrier for protein delivery.(28) Xanthan gum has been incorporated in an ophthalmic liquid dosage form, which interacts with mucin, thereby helping in the prolonged retention of the dosage form in the precorneal area.(29) When added to liquid ophthalmics, xanthan gum delays the release of active substances, increasing the therapeutic activity of the pharmaceutical formulations.(30) Xanthan gum can be used to increase the bioadhesive strength in vaginal formulations.(31) Xanthan gum alone or with carbopol 974P has been used as a mucoadhesive controlled-release excipient for buccal drug delivery.(32,33) Modified xanthan films have been used as a matrix system for transdermal delivery of atenolol.(34) Xanthan gum has also been used as a gelling agent for topical formulations incorporating solid lipid nanoparticles of vitamin A(35) or microemulsion of ibuprofen.(36) A combined polymer system consisting of xanthan gum, carboxy methylcellulose and a polyvinyl pyrolidone backboned polymer has been used for relieving the symptoms of xerostomia.(37) Xanthan gum can also be used as an excipient for spray-drying and freeze-drying processes for better results.(38,39) It has been successfully used alone or in combination with agar for microbial culture media.(40) Xanthan gum is also used as a hydrocolloid in the food industry, and in cosmetics it has been used as a thickening agent in shampoo.(41) Polyphosphate with xanthum gum in soft drinks is suggested to be effective at reducing erosion of enamel.(42,43
Xanthan gum occurs as a cream- or white-colored, odorless, freeflowing, fine powder.
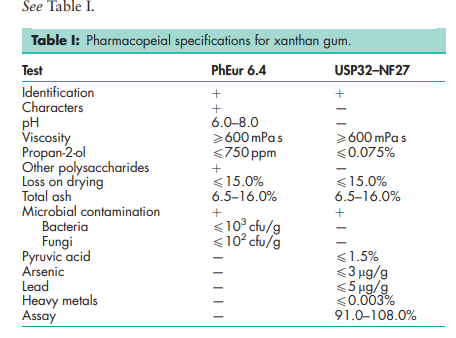
See Table I.
Acidity/alkalinity pH = 6.0–8.0 for a 1% w/v aqueous solution. Freezing point 08C for a 1% w/v aqueous solution. Heat of combustion 14.6 J/g (3.5 cal/g) Melting point Chars at 2708C. NIR spectra see Figure 1. Particle size distribution Various grades with different particle sizes are available; see Table II. Refractive index nD 20 = 1.333 (1% w/v aqueous solution). Solubility Practically insoluble in ethanol and ether; soluble in cold or warm water. Specific gravity 1.600 at 258C Viscosity (dynamic) 1200–1600 mPa s (1200–1600 cP) for a 1% w/v aqueous solution at 258C.
Xanthan gum is a stable material. Aqueous solutions are stable over a wide pH range (pH 3–12), although they demonstrate maximum stability at pH 4–10 and temperatures of 10–608C. Xanthan gum solutions of less than 1% w/v concentration may be adversely affected by higher than ambient temperatures: for example, viscosity is reduced. Xanthan gum provides the same thickening, stabilizing, and suspending properties during long-term storage at elevated temperatures as it does at ambient conditions. In addition, it ensures excellent freeze–thaw stability. Solutions are also stable in the presence of enzymes, salts, acids, and bases. Vanzan NF-ST is especially designed for use in systems containing high salt concentrations as it dissolves directly in salt solutions, and its viscosity is relatively unaffected by high salt levels as compared with general purpose grades. The bulk material should be stored in a well-closed container in a cool, dry place.
Xanthan gum is an anionic material and is not usually compatible with cationic surfactants, polymers, or preservatives, as precipitation occurs. Anionic and amphoteric surfactants at concentrations above 15% w/v cause precipitation of xanthan gum from a solution. Under highly alkaline conditions, polyvalent metal ions such as calcium cause gelation or precipitation; this may be inhibited by the addition of a glucoheptonate sequestrant. The presence of low levels of borates (<300 ppm) can also cause gelation. This may be avoided by increasing the boron ion concentration or by lowering the pH of a formulation to less than pH 5. The addition of ethylene glycol, sorbitol, or mannitol may also prevent this gelation. Xanthan gum is compatible with most synthetic and natural viscosity-increasing agents, many strong mineral acids, and up to 30% inorganic salts. If it is to be combined with cellulose derivatives, then xanthan gum free of cellulase should be used to prevent depolymerization of the cellulose derivative. Xanthan gum solutions are stable in the presence of up to 60% water-miscible organic solvents such as acetone, methanol, ethanol, or propan-2- ol. However, above this concentration precipitation or gelation occurs. The viscosity of xanthan gum solutions is considerably increased, or gelation occurs, in the presence of some materials such as ceratonia, guar gum, and magnesium aluminum silicate.(9) This effect is most pronounced in deionized water and is reduced by the presence of salt. This interaction may be desirable in some instances and can be exploited to reduce the amount of xanthan gum used in a formulation; see Section 7. Xanthan gum is incompatible with oxidizing agents, some tablet film-coatings,(4) carboxymethylcellulose sodium,(44) dried aluminum hydroxide gel,(45) and some active ingredients such as amitriptyline, tamoxifen, and verapamil.(3)
Xanthan gum is a polysaccharide produced by a pure-culture aerobic fermentation of a carbohydrate with Xanthomonas campestris. The polysaccharide is then purified by recovery with propan-2-ol, dried, and milled.(.(46,47)
Xanthan gum is widely used in oral and topical pharmaceutical formulations, cosmetics, and food products, and is generally regarded as nontoxic and nonirritant at the levels employed as a pharmaceutical excipient. The estimated acceptable daily intake for xanthan gum has been set by the WHO at up to 10 mg/kg body-weight.(48) No eye or skin irritation has been observed in rabbits and no skin allergy has been observed in guinea pigs following skin exposure. No adverse effects were observed in long term feeding studies with rats (up to 1000 mg/kg/day) and dogs (up to 1000 mg/kg/day). No adverse effects were observed in a three-generation reproduction study with rats (up to 500 mg/kg/day).(49) LD50 (dog, oral): >20 g/kg(48) LD50 (rat, oral): >45 g/kg LD50 (mouse, oral): >1 g/kg(50) LD50 (mouse, IP): >50 mg/kg(50) LD50 (mouse, IV): 100–250 mg/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (oral solutions, suspensions, and tablets; rectal and topical preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients
Ceratonia; guar gum.
Xanthan gum is available in several different grades that have varying particle sizes. Fine-mesh grades of xanthan gum are used in applications where high solubility is desirable since they dissolve rapidly in water. However, fine-mesh grades disperse more slowly than coarse grades and are best used dry blended with the other ingredients of a formulation. In general, it is preferable to dissolve xanthan gum in water first and then add the other ingredients of a formulation. Novel pH-sensitive hydrogel beads have been prepared using a copolymer of poly(acrylamide-g-xanthan) for targeting ketoprofen to the intestine.(51) These beads were able to retard drug release in the stomach, thus diminshing gastric side effects such as ulceration, hemorrhage and erosion of gastric mucosa.(51) Bioadhesive nasal inserts prepared from xanthan gum have a high potential as a new nasal dosage form for extended drug delivery.(52) Xanthan gum wafers have potential as drug delivery systems for suppurating wounds.(53,54) The USP32–NF27 also includes a monograph for xanthan gum solution. A specification for xanthan gum is contained in the Food Chemicals Codex (FCC).(55) The EINECS number for xanthan gum is 234-394-2. The PubChem Compound ID (CID) for xanthan gum is 7107.