

Name: Albumin
CAS No: Serum albumin [9048-49-1]
BP: Albumin Solution PhEur: Human Albumin Solution USP: Albumin Human
Alba; Albuconn; Albuminar; albumin human solution; albumini humani solutio; Albumisol; Albuspan; Albutein; Buminate; human serum albumin; normal human serum albumin; Octalbin; Plasbumin; plasma albumin; Pro-Bumin; Proserum; Zenalb.
Serum albumin [9048-49-1]
Human serum albumin has a molecular weight of about 66 500 and is a single polypeptide chain consisting of 585 amino acids.
Primary structure Human albumin is a single polypeptide chain of 585 amino acids and contains seven disulfide bridges. Secondary structure Human albumin is known to have a secondary structure that is about 55% a-helix. The remaining 45% is believed to be divided among turns, disordered, and b structures.(1) Albumin is the only major plasma protein that does not contain carbohydrate constituents. Assays of crystalline albumin show less than one sugar residue per molecule.
Albumin is primarily used as an excipient in parenteral pharmaceutical formulations, where it is used as a stabilizing agent for formulations containing proteins and enzymes.(2) Albumin has also been used to prepare microspheres and microcapsules for experimental drug-delivery systems.(3–6) As a stabilizing agent, albumin has been employed in protein formulations at concentrations as low as 0.003%, although concentrations of 1–5% are commonly used. Albumin has also been used as a cosolvent(7) for parenteral drugs, as a cryoprotectant during lyophilization,(8,9) and to prevent adsorption of other proteins to surfaces. Therapeutically, albumin solutions have been used parenterally for plasma volume replacement and to treat severe acute albumin loss. However, the benefits of using albumin in such applications in critically ill patients has been questioned.(10)
The USP 32 describes albumin human as a sterile nonpyrogenic preparation of serum albumin obtained from healthy human donors; see Section 13. It is available as a solution containing 4, 5, 20, or 25 g of serum albumin in 100 mL of solution, with not less than 96% of the total protein content as albumin. The solution contains no added antimicrobial preservative but may contain sodium acetyltryptophanate with or without sodium caprylate as a stablizing agent. The PhEur 6.0 similarly describes albumin solution as an aqueous solution of protein obtained from human plasma; see Section 13. It is available as a concentrated solution containing 150–250 g/L of total protein or as an isotonic solution containing 35–50 g/L of total protein. Not less than 95% of the total protein content is albumin. A suitable stabilizer against the effects of heat, such as sodium caprylate (sodium octanoate) or N-acetyltryptophan or a combination of these two at a suitable concentration, may be added, but no antimicrobial preservative is added. Aqueous albumin solutions are slightly viscous and range in color from almost colorless to amber depending upon the protein concentration. In the solid state, albumin appears as brownish amorphous lumps, scales, or powder.
See Table I.
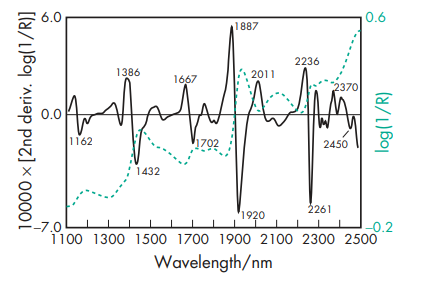
Acidity/alkalinity pH = 6.7–7.3 for a 1% w/v solution, in 0.9% w/v sodium chloride solution, at 208C. NIR spectra see Figure 1. Osmolarity A 4–5% w/v aqueous solution is isoosmotic with serum. Solubility Freely soluble in dilute salt solutions and water. Aqueous solutions containing 40% w/v albumin can be readily prepared at pH 7.4. The high net charge of the peptide contributes to its solubility in aqueous media. The seven disulfide bridges contribute to its chemical and spatial conformation. At physiological pH, albumin has a net electrostatic charge of about –17. Aqueous albumin solutions are slightly viscous and range in color from almost colorless to amber depending on the protein concentration.
Albumin is a protein and is therefore susceptible to chemical degradation and denaturation by exposure to extremes of pH, high salt concentrations, heat, enzymes, organic solvents, and other chemical agents. Albumin solutions should be protected from light and stored at a temperature of 2–258C or as indicated on the label.
See Section 11
Albumin human (USP 32) Albumin human is a sterile nonpyrogenic preparation of serum albumin that is obtained by fractionating material (source blood, plasma, serum, or placentas) from healthy human donors. The source material is tested for the absence of hepatitis B surface antigen. It is made by a process that yields a product safe for intravenous use. Human albumin solution (PhEur 6.0) Human albumin solution is an aqueous solution of protein obtained from plasma. Separation of the albumin is carried out under controlled conditions so that the final product contains not less than 95% albumin. Human albumin solution is prepared as a concentrated solution containing 150–250 g/L of total protein or as an isotonic solution containing 35–50 g/L of total protein. A suitable stabilizer against the effects of heat such as sodium caprylate (sodium octanoate) or N-acetyltryptophan or a combination of these two at a suitable concentration, may be added, but no antimicrobial preservative is added at any stage during preparation. The solution is passed through a bacteriaretentive filter and distributed aseptically into sterile containers,
Albumin occurs naturally in the body, comprising about 60% of all the plasma proteins. As an excipient, albumin is used primarily in parenteral formulations and is generally regarded as an essentially nontoxic and nonirritant material. Adverse reactions to albumin infusion rarely occur but include nausea, vomiting, increased salivation, chills, and febrile reactions. Urticaria and skin rash have been reported. Allergic reactions, including anaphylactic shock, can occur. Albumin infusions are contraindicated in patients with severe anemia or cardiac failure. Albumin solutions with aluminum content of less than 200 mg/L should be used in dialysis patients and premature infants.(11) LD50 (monkey, IV): >12.5 g/kg(12) LD50 (rat, IV): >12.5 g/kg
Observe handling precautions appropriate for a biologically derived blood product.
Included in the FDA Inactive Ingredients Database (oral, tablets, film-coatings; IV injections, IV infusions and subcutaneous injectables). Included in parenteral products licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Albumins derived from animal sources are also commercially available, e.g. bovine serum albumin.
A 100 mL aqueous solution of albumin containing 25 g of serum albumin is osmotically equivalent to 500 mL of normal human plasma. The EINECS number for albumin is 310-127-6.