

Name: Xylitol
CAS No: xylo-Pentane-1,2,3,4,5-pentol [87-99-0]
BP: Xylitol JP: Xylitol PhEur: Xylitol USP-NF: Xylitol
E967; Klinit; meso-xylitol; xilitol; Xylifin; Xylisorb; xylit; Xylitab; xylite; Xylitolo; xylitolum.
xylo-Pentane-1,2,3,4,5-pentol [87-99-0]
C5H12O5 , 152.15
Xylitol is used as a noncariogenic sweetening agent in a variety of pharmaceutical dosage forms, including tablets, syrups, and coatings. It is also widely used as an alternative to sucrose in foods and as a base for medicated confectionery. Xylitol is finding increasing application in chewing gum,(1,2) mouthrinses,(3) and toothpastes(4) as an agent that decreases dental plaque and tooth decay (dental caries). Unlike sucrose, xylitol is not fermented into cariogenic acid end products(5) and it has been shown to reduce dental caries by inhibiting the growth of cariogenic Streptococcus mutans bacteria.(6,7) As xylitol has an equal sweetness intensity to sucrose, combined with a distinct cooling effect upon dissolution of the crystal, it is highly effective in enhancing the flavor of tablets and syrups and masking the unpleasant or bitter flavors associated with some pharmaceutical actives and excipients. In topical cosmetic and toiletry applications, xylitol is used primarily for its humectant and emollient properties, although it has also been reported to enhance product stability through a combination of potentiation of preservatives and its own bacteriostatic and bactericidal properties. Granulates of xylitol are used as diluents in tablet formulations, where they can provide chewable tablets with a desirable sweet taste and cooling sensation, without the ‘chalky’ texture experienced with some other tablet diluents. Xylitol solutions are employed in tablet-coating applications at concentrations in excess of 65% w/w. Xylitol coatings are stable and provide a sweet-tasting and durable hard coating. In liquid preparations, xylitol is used as a sweetening agent and vehicle for sugar-free formulations. In syrups, it has a reduced tendency to ‘cap-lock’ by effectively preventing crystallization around the closures of bottles. Xylitol also has a lower water activity and a higher osmotic pressure than sucrose, therefore enhancing product stability and freshness. In addition, xylitol has also been demonstrated to exert certain specific bacteriostatic and bactericidal effects, particularly against common spoilage organisms.(8,9) Therapeutically, xylitol is additionally utilized as an energy source for intravenous infusion therapy following trauma.(10
Xylitol occurs as a white, granular solid comprising crystalline, equidimensional particles having a mean diameter of about 0.4–0.6 mm. It is odorless, with a sweet taste that imparts a cooling sensation. Xylitol is also commercially available in powdered form, and several granular, directly compressible forms.(11)
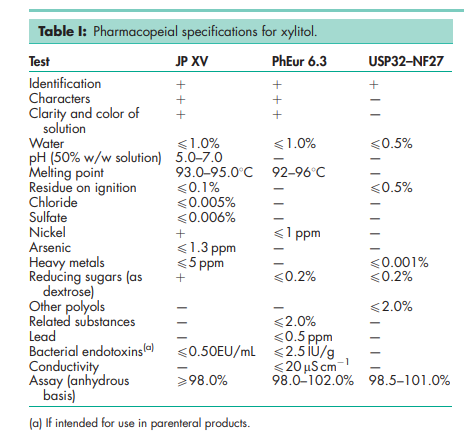
See Table I.
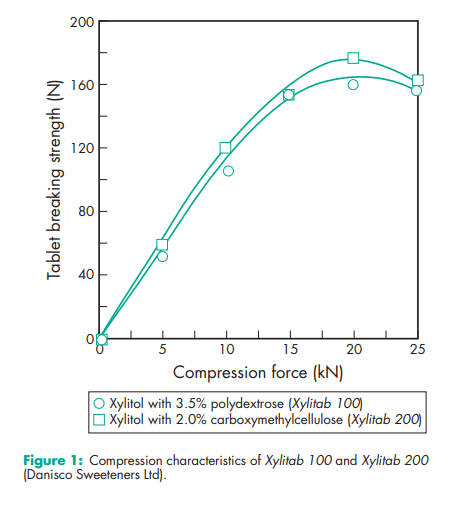
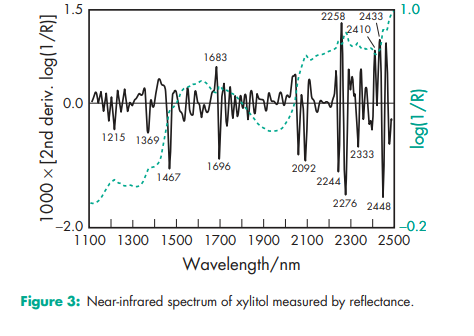
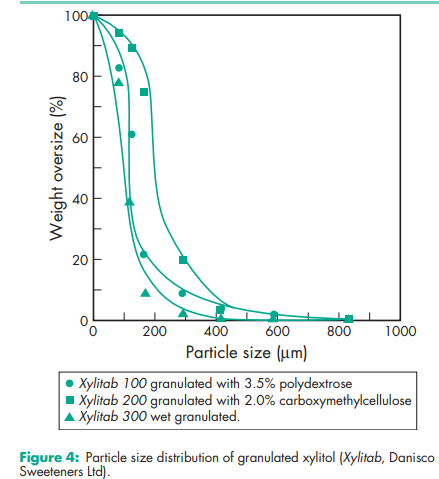
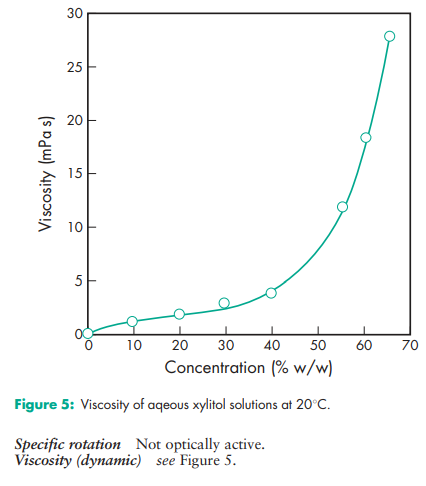
Acidity/alkalinity pH = 5.0–7.0 (10% w/v aqueous solution). Boiling point 215–2178C Compressibility see Figure 1. Crystalline xylitol, under the same test conditions as illustrated in Figure 1, produces 12.5 mm tablets of 40 N hardness at 20 kN compression force. Density (true) 1.52 g/cm3 Density (bulk) 0.8–0.85 g/cm3 for crystalline xylitol; 0.5–0.7 g/cm3 for directly compressible granulated grades Flowability Flow characteristics vary depending upon the particle size of xylitol used. Fine-milled grades tend to be relatively poorly flowing, while granulated grades have good flow properties. Heat of solution 157.1 kJ/kg (–36.7 cal/g) Melting point 92.0–96.08C Moisture content Xylitol is a moderately hygroscopic powder under normal conditions; see also Figure 2. At 208C and 52% relative humidity, the equilibrium moisture content of xylitol is 0.1% w/w. After drying in a vacuum, over P2O5 at 808C for 4 hours, xylitol loses less than 0.5% w/w water. NIR spectra see Figure 3. Osmolarity A 4.56% w/v aqueous solution is iso-osmotic with serum. Particle size distribution The particle size distribution of xylitol depends upon the grade selected. Normal crystalline material typically has a mean particle size of 0.4–0.6 mm. Milled grades are commercially available that offer mean particle sizes as low as 50 mm. Individual suppliers’ literature should be consulted for further information. For particle size distributions of granulated xylitol, see Figure 4. Solubility see Table II

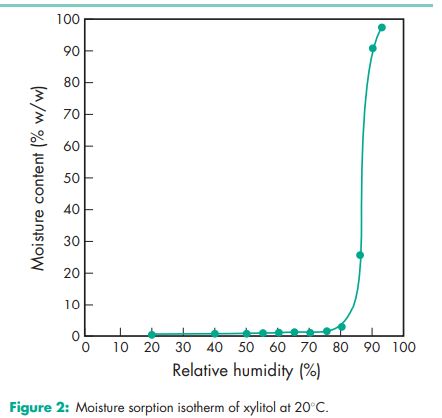

Xylitol is stable to heat but is marginally hygroscopic. Caramelization can occur only if it is heated for several minutes near its boiling point. Crystalline material is stable for at least 3 years if stored at less than 65% relative humidity and 258C. Milled and specialized granulated grades of xylitol have a tendency to cake and should therefore be used within 9 to 12 months. Aqueous xylitol solutions have been reported to be stable, even on prolonged heating and storage. Since xylitol is not utilized by most microorganisms, products made with xylitol are usually safe from fermentation and microbial spoilage.(8,9) Xylitol should be stored in a well-closed container in a cool, dry place.
Xylitol is incompatible with oxidizing agents.
Xylitol occurs naturally in many fruits and berries, although extraction from such sources is not considered to be commercially viable. Industrially, xylitol is most commonly derived from various types of hemicellulose obtained from such sources as wood, corn cobs, cane pulp, seed hulls, and shells. These materials typically contain 20–35% xylan, which is readily converted to xylose (wood sugar) by hydrolysis. This xylose is subsequently converted to xylitol via hydrogenation (reduction). Following the hydrogenation step, there are a number of separation and purification steps that ultimately yield high-purity xylitol crystals. The nature of this process, and the stringent purification procedures employed, result in a finished product with a very low impurity content. Potential impurities that may appear in small quantities are mannitol, sorbitol, galactitol, or arabitol. Less commonly employed methods of xylitol manufacture include the conversion of glucose (dextrose) to xylose followed by hydrogenation to xylitol, and the microbiological conversion of xylose to xylitol.
Xylitol is used in oral pharmaceutical formulations, confectionery, and food products, and is generally regarded as an essentially nontoxic, nonallergenic, and nonirritant material. Xylitol has an extremely low relative glycemic response and is metabolized independently of insulin. Following ingestion of xylitol, the blood glucose and serum insulin responses are significantly lower than following ingestion of glucose or sucrose. These factors make xylitol a suitable sweetener for use in diabetic or carbohydrate-controlled diets.(12) Up to 100 g of xylitol in divided oral doses may be tolerated daily, although, as with other polyols, large doses may have a laxative effect. The laxative threshold depends on a number of factors, including individual sensitivity, mode of ingestion, daily diet, and previous adaptation to xylitol. Single doses of 20–30 g and daily doses of 0.5–1.0 g/kg body-weight are usually well tolerated by most individuals. Approximately 25–50% of the ingested xylitol is absorbed, with the remaining 50–75% passing to the lower gut, where it undergoes indirect metabolism via fermentative degradation by the intestinal flora. An acceptable daily intake for xylitol of ‘not specified’ has been set by the WHO since the levels used in foods do not represent a hazard to health.(13) LD50 (mouse, IP): 22.1 g/kg(14,15) LD50 (mouse, IV): 12 g/kg LD50 (mouse, oral): 12.5 g/kg LD50 (rat, oral): 17.3 g/kg LD50 (rat, IV): 10.8 g/kg LD50 (rabbit, oral): 16.5 g/kg LD50 (rabbit, IV): 4 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Xylitol may cause transient gastrointestinal discomfort if ingested in large quantities; and may also be irritant to the eyes. Eye protection and gloves are recommended.Conventional dust-control practices should be employed. Xylitol is flammable, but does not ignite readily.
GRAS listed. Approved for use as a food additive in over 70 countries worldwide, including Europe, the USA and Japan. Included in the FDA Inactive Ingredients Database (oral solution, chewing gum). Included in nonparenteral medicines licensed in the UK and USA. Included in the Canadian List of Acceptable Nonmedicinal Ingredients
Various directly compressible forms of xylitol that contain other excipients are commercially available, e.g. Xylitab 100, which contains 3.5% polydextrose, and Xylitab 200, which contains 2.0% carboxymethylcellulose (both Danisco Sweeteners Ltd). A directly compressible form of pure xylitol is also available, Xylitab 300 (Danisco Sweeteners Ltd), which is produced via wet granulation. Pyrogen-free grades of xylitol suitable for parenteral use are also commercially available.
The sweetening power of xylitol is approximately equal to that of sucrose, although it has been shown to be pH-, concentration-, and temperature-dependent; xylitol is approximately 2.5 times as sweet as mannitol. Xylitol is highly chemically stable, meaning that it will not interact with pharmaceutical actives or excipients, and can be utilized over a wide pH range (pH 1–11). Xylitol has a negative heat of solution that is far larger than that of other alternative sweetening agents; see Table III. Because of this, xylitol produces an intense cooling effect as the crystalline material dissolves. Xylitol’s combination of sweetness and cooling can create product appeal while helping to mask the undesirable taste of many pharmaceutical actives or excipients. A specification for xylitol is contained in the Food Chemicals Codex (FCC).(16) The EINECS number for xylitol is 201-788-0. The PubChem Compound ID (CID) for xylitol is 6912.
