

Name: Cottonseed Oil
CAS No: Cottonseed oil [8001-29-4]
USP-NF: Cottonseed Oil
Cotton oil; refined cottonseed oil.
Cottonseed oil [8001-29-4]
A typical analysis of refined cottonseed oil indicates the composition of the acids present as glycerides to be as follows: linoleic acid 39.3%; oleic acid 33.1%; palmitic acid 19.1%; stearic acid 1.9%; arachidic acid 0.6%, and myristic acid 0.3%. Also present are small quantities of phospholipid, phytosterols, and pigments. The toxic polyphenolic pigment gossypol is present in raw cottonseed and in the oil cake remaining after expression of oil; it is not found in the refined oil
See Section 4.
Cottonseed oil is used in pharmaceutical formulations primarily as a solvent for intramuscular injections. It has been used in intravenous emulsions as a fat source in parenteral nutrition regimens, although its use for this purpose has been superseded by soybean oil emulsions; see Section 14. It has also been used as an adjuvant in cholecystography and as a pediculicide and acaricide. It has the nutritive and emollient properties of fixed vegetable oils. By virtue of its high content of unsaturated acid glycerides (especially linoleic acid), it is used for dietary control of blood cholesterol levels in the prophylaxis and treatment of atherosclerosis. It is used as a solvent and vehicle for injections; as an emollient vehicle for other medications; and orally as a mild cathartic (in a dose of 30 mL or more). It can also retard gastric secretion and motility, and increase caloric intake. It has been used in the manufacture of soaps, oleomargarine, lard substitutes, glycerin, lubricants, and cosmetics. Cottonseed oil has been used as a tablet binder for acetaminophen; for characterization of the hot-melt fluid bed coating process;(1) in the manufacturing of stable oral pharmaceutical powders; in encapsulation of enzymes; and as an aqueous dispersion in pharmaceutical coating
Pale yellow or bright golden yellow-colored, clear oily liquid. It is odorless, or nearly so, with a bland, nutty taste. At temperatures below 108C particles of solid fat may separate from the oil, and at about –5 to 08C the oil becomes solid or nearly so. If it solidifies, the oil should be remelted and thoroughly mixed before use
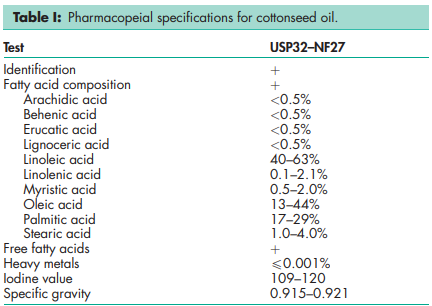
See Table I.
Autoignition temperature 3448C Density 0.916 g/cm3 Flash point 3218C Freezing point 5 to 08C Heat of combustion 37.1 kJ/g Refractive index n D 40 = 1.4645–1.4655 Solubility Slightly soluble in ethanol (95%); miscible with carbon disulfide, chloroform, ether, hexane, and petroleum ether. Surface tension 35.4 mN/m (35.4 dynes/cm) at 208C; 31.3 mN/m (31.3 dynes/cm) at 808C. Viscosity (dynamic) Up to 70.4 mPa s (70.4 cP) at 208C
Cottonseed oil is stable if stored in a well-filled, airtight, lightresistant container in a cool, dry place. Avoid exposure to excessive heat.
Cottonseed oil is the refined fixed oil obtained from the seed of cultivated varieties of Gossypium hirsutum Linne´ or of other species of Gossypium (Fam. Malvaceae). The seeds contain about 15% oil. The testae of the seeds are first separated and the kernels are then exposed to powerful expression in a hydraulic press. The crude oil thus obtained has a bright red or blackish-red color and requires purification before it is suitable for food or pharmaceutical purposes. Cottonseed oil is refined by treatment with diluted alkali to neutralize acids, decolorized with fuller’s earth or activated carbon, deodorized with steam under reduced pressure, and chilled to separate glycerides and resinous substances of higher melting point.
Cottonseed oil emulsions have in the past been used in long-term intravenous nutrition regimens.(2,3) A complex of adverse reactions,called the ‘overloading syndrome’ (4) has been seen with chronic administration of cottonseed oil emulsion. This consisted of anorexia, nausea, abdominal pain, headache, fever, and sore throat. Signs of impaired liver function, anemia, hepatosplenomegaly, thrombocytopenia, and spontaneous hemorrhage due to delayed blood clotting have been reported. For parenteral nutrition purposes, cottonseed oil has been replaced by soybean oil,(2,5,6) especially in pregnant women, where the use of cottonseed lipid emulsion has been associated with adverse effects.(7) A notable difference between the cottonseed oil emulsion and the soybean oil emulsion is the particle size. The cottonseed oil emulsion has much larger particles than the soybean oil emulsion. These larger particles may have been handled differently by the body, thus perhaps accounting for some of the toxic reactions.
Observe normal precautions appropriate to the circumstances and quantity of material handled. Spillages of this material are very slippery and should be covered with an inert absorbent material prior to disposal. Cottonseed oil is a combustible liquid when exposed to heat or flame. If it is allowed to impregnate rags or oily waste, there is a risk due to spontaneous heating. Dry chemicals such as carbon dioxide should be used to fight any fires.
Included in the FDA Inactive Ingredients Database (IM injections, oral, capsule, tablet and sublingual preparations). Included in the Canadian List of Acceptable Non-medicinal Ingredients
Almond oil; canola oil; corn oil; hydrogenated vegetable oil; peanut oil; sesame oil; soybean oil; sunflower oil.
The USP32–NF27, PhEur 6.2, and BP 2009 also list hydrogenated cottonseed oil. A specification for unhydrogenated cottonseed oil is contained in the Food Chemicals Codex (FCC).(8) The EINECS number for cottonseed oil is 232-280-7.