

Name: Croscarmellose Sodium
CAS No: Cellulose, carboxymethyl ether, sodium salt, crosslinked [74811-65-7]
BP: Croscarmellose Sodium JP: Croscarmellose Sodium PhEur: Croscarmellose Sodium USP-NF: Croscarmellose Sodium
Ac-Di-Sol; carmellosum natricum conexum; crosslinked carboxymethylcellulose sodium; Explocel; modified cellulose gum; Nymcel ZSX; Pharmacel XL; Primellose; Solutab; Vivasol.
Cellulose, carboxymethyl ether, sodium salt, crosslinked [74811-65-7]
Croscarmellose sodium is a crosslinked polymer of carboxymethylcellulose sodium. See Carboxymethylcellulose sodium.
See Carboxymethylcellulose sodium
Croscarmellose sodium is used in oral pharmaceutical formulations as a disintegrant for capsules,(1,2) tablets,(3–13) and granules. In tablet formulations, croscarmellose sodium may be used in both direct-compression and wet-granulation processes. When used in wet granulations, the croscarmellose sodium should be added in both the wet and dry stages of the process (intra- and extragranularly) so that the wicking and swelling ability of the disintegrant is best utilized.(11,12) Croscarmellose sodium at concentrations up to 5% w/w may be used as a tablet disintegrant, although normally 2% w/w is used in tablets prepared by direct compression and 3% w/w in tablets prepared by a wet-granulation process. See Table I.
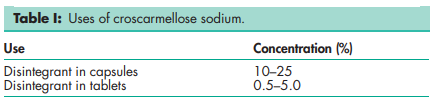
Croscarmellose sodium occurs as an odorless, white or grayishwhite powder.
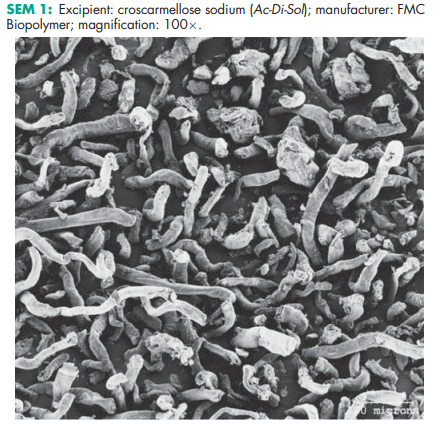
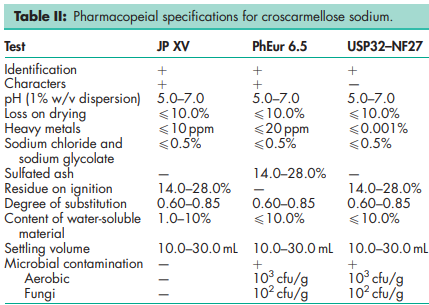
See Table II
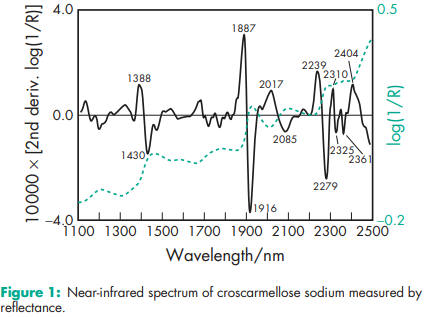
Acidity/alkalinity pH = 5.0–7.0 in aqueous dispersions. Bonding index 0.0456 Brittle fracture index 0.1000 Density (bulk) 0.529 g/cm3 for Ac-Di-Sol Density (tapped) 0.819 g/cm3 for Ac-Di-Sol Density (true) 1.543 g/cm3 for Ac-Di-Sol NIR spectra see Figure 1. Particle size distribution Ac-Di-Sol: not more than 2% retained on a #200 (73.7 mm) mesh and not more than 10% retained on a #325 (44.5 mm) mesh. Solubility Insoluble in water, although croscarmellose sodium rapidly swells to 4–8 times its original volume on contact with water. Practically insoluble in acetone, ethanol and toluene. Specific surface area 0.81–0.83 m2 /g
Croscarmellose sodium is a stable though hygroscopic material. A model tablet formulation prepared by direct compression, with croscarmellose sodium as a disintegrant, showed no significant difference in drug dissolution after storage at 308C for 14 months.(9) Croscarmellose sodium should be stored in a well-closed container in a cool, dry place.
The efficacy of disintegrants, such as croscarmellose sodium, may be slightly reduced in tablet formulations prepared by either the wet-granulation or direct-compression process that contain hygroscopic excipients such as sorbitol.(10) Croscarmellose sodium is not compatible with strong acids or with soluble salts of iron and some other metals such as aluminum, mercury, and zinc.
Alkali cellulose is prepared by steeping cellulose, obtained from wood pulp or cotton fibers, in sodium hydroxide solution. The alkali cellulose is then reacted with sodium monochloroacetate to obtain carboxymethylcellulose sodium. After the substitution reaction is completed and all of the sodium hydroxide has been used, the excess sodium monochloroacetate slowly hydrolyzes to glycolic acid. The glycolic acid changes a few of the sodium carboxymethyl groups to the free acid and catalyzes the formation of crosslinks to produce croscarmellose sodium. The croscarmellose sodium is then extracted with aqueous alcohol and any remaining sodium chloride or sodium glycolate is removed. After purification, croscarmellose sodium of purity greater than 99.5% is obtained.(4) The croscarmellose sodium may be milled to break the polymer fibers into shorter lengths and hence improve its flow properties
Croscarmellose sodium is mainly used as a disintegrant in oral pharmaceutical formulations and is generally regarded as an essentially nontoxic and nonirritant material. However, oral consumption of large amounts of croscarmellose sodium may have a laxative effect, although the quantities used in solid dosage formulations are unlikely to cause such problems. In the UK, croscarmellose sodium is accepted for use in dietary supplements. The WHO has not specified an acceptable daily intake for the related substance carboxymethylcellulose sodium, used as a food additive, since the levels necessary to achieve a desired effect were not considered sufficient to be a hazard to health.(14) See also Carboxymethylcellulose Sodium.
Observe normal precautions appropriate to the circumstances and quantity of material handled. Croscarmellose sodium may be irritant to the eyes; eye protection is recommended.
Included in the FDA Inactive Ingredients Database (oral capsules, granules, sublingual tablets, and tablets). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Carboxymethylcellulose calcium; carboxymethylcellulose sodium.
Croscarmellose sodium is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. Typically, the degree of substitution (DS) for croscarmellose sodium is 0.7. The EINECs number for croscarmellose sodium is 232-674-9