

Name: Crospovidone
CAS No: 1-Ethenyl-2-pyrrolidinone homopolymer [9003-39-8]
BP: Crospovidone PhEur: Crospovidone USP-NF: Crospovidone
Crospovidonum; Crospopharm; crosslinked povidone; E1202; Kollidon CL; Kollidon CL-M; Polyplasdone XL; Polyplasdone XL-10; polyvinylpolypyrrolidone; PVPP; 1-vinyl-2-pyrrolidinone homopolymer.
1-Ethenyl-2-pyrrolidinone homopolymer [9003-39-8]
(C6H9NO)n >1 000 000 The USP32–NF27 describes crospovidone as a water-insoluble synthetic crosslinked homopolymer of N-vinyl-2-pyrrolidinone. An exact determination of the molecular weight has not been established because of the insolubility of the material.
See Povidone
Crospovidone is a water-insoluble tablet disintegrant and dissolution agent used at 2–5% concentration in tablets prepared by directcompression or wet- and dry-granulation methods.(1–6) It rapidly exhibits high capillary activity and pronounced hydration capacity, with little tendency to form gels. Studies suggest that the particle size of crospovidone strongly influences disintegration of analgesic tablets.(7) Larger particles provide a faster disintegration than smaller particles. Crospovidone can also be used as a solubility enhancer. With the technique of co-evaporation, crospovidone can be used to enhance the solubility of poorly soluble drugs. The drug is adsorbed on to crospovidone in the presence of a suitable solvent and the solvent is then evaporated. This technique results in faster dissolution rate.
Crospovidone is a white to creamy-white, finely divided, freeflowing, practically tasteless, odorless or nearly odorless, hygroscopic powder.
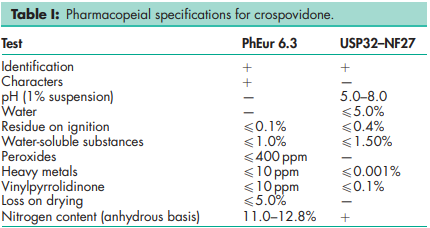
See Table I
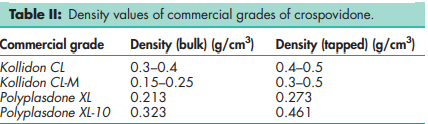
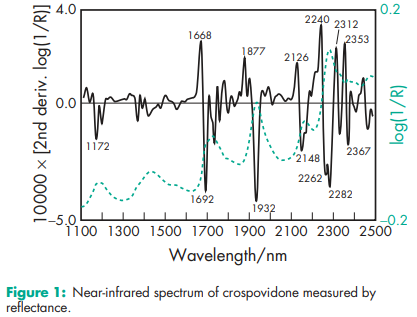
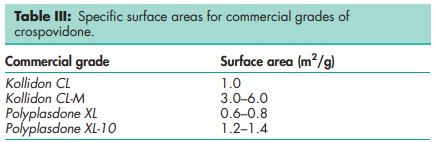
Acidity/alkalinity pH = 5.0–8.0 (1% w/v aqueous slurry) Density 1.22 g/cm3 Density (bulk) see Table II. Density (tapped) see Table II. Moisture content Maximum moisture sorption is approximately 60%. NIR spectra see Figure 1. Particle size distribution Less than 400 mm for Polyplasdone XL; less than 74 mm for Polyplasdone XL-10. Approximately 50% greater than 50 mm and maximum of 3% greater than 250 mm in size for Kollidon CL. Minimum of 90% of particles are below 15 mm for Kollidon CL-M. The average particle size for Crospopharm type A is 100 mm and for Crospopharm type B it is 30 mm. Solubility Practically insoluble in water and most common organic solvents. Specific surface area

Since crospovidone is hygroscopic, it should be stored in an airtight container in a cool, dry place
Crospovidone is compatible with most organic and inorganic pharmaceutical ingredients. When exposed to a high water level, crospovidone may form molecular adducts with some materials; see Povidone
Acetylene and formaldehyde are reacted in the presence of a highly active catalyst to form butynediol, which is hydrogenated to butanediol and then cyclodehydrogenated to form butyrolactone. Pyrrolidone is produced by reacting butyrolactone with ammonia. This is followed by a vinylation reaction in which pyrrolidone and acetylene are reacted under pressure. The monomer vinylpyrrolidone is then polymerized in solution, using a catalyst. Crospovidone is prepared by a ‘popcorn polymerization’ process
Crospovidone is used in oral pharmaceutical formulations and is generally regarded as a nontoxic and nonirritant material. Shortterm animal toxicity studies have shown no adverse effects associated with crospovidone.(8) However, owing to the lack of available data, an acceptable daily intake in humans has not been specified by the WHO.(8) LD50 (mouse, IP): 12 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection, gloves, and a dust mask are recommended
Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (IM injections, oral capsules and tablets; topical, transdermal, and vaginal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients
Copovidone; povidone.
Crospovidone is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. Crospovidone has been studied as a superdisintegrant. The ability of the compound to swell has been examined directly using scanning electron microscopy.(9) The impact of crospovidone on percolation has also been examined.(10) The impact of crospovidone on dissolution of poorly soluble drugs in tablets has also been investigated.(11) Crospovidone has been shown to be effective with highly hygroscopic drugs.(12) It continues to be examined for its uses in a number of tablet formulations. A specification for crospovidone is contained in the Food Chemicals Codex (FCC).(13) The PubChem Compound ID (CID) for crospovidone is 6917.