

Name: Cyclodextrins
CAS No: a-Cyclodextrin [10016-20-3], b-Cyclodextrin [7585-39-9], g-Cyclodextrin [17465-86-0]
BP: Alfadex Betadex PhEur: Alfadex Betadex USP-NF: Alfadex Betadex Gamma Cyclodextrin
Cyclodextrin Cavitron; cyclic oligosaccharide; cycloamylose; cycloglucan; Encapsin; Schardinger dextrin. a-Cyclodextrin alfadexum; alpha-cycloamylose; alpha-cyclodextrin; alpha-dextrin; Cavamax W6 Pharma; cyclohexaamylose; cyclomaltohexose. b-Cyclodextrin beta-cycloamylose; beta-dextrin; betadexum; Cavamax W7 Pharma; cycloheptaamylose; cycloheptaglucan; cyclomaltoheptose; Kleptose. g-Cyclodextrin Cavamax W8 Pharma; cyclooctaamylose; cyclomaltooctaose.
a-Cyclodextrin [10016-20-3], b-Cyclodextrin [7585-39-9], g-Cyclodextrin [17465-86-0]
a-Cyclodextrin C36H60O30 972 b-Cyclodextrin C42H70O35 1135 g-Cyclodextrin C48H80O40 1297
Note: the structure of betadex (b-cyclodextrin) with 7 glucose units is shown. R = H for ‘natural’ a, b, and g-cyclodextrins with 6, 7 and 8 glucose units, respectively R = H or CH3 for methyl cyclodextrins R = H or CHOHCH3 for 2-hydroxyethyl cyclodextrins R = H or CH2CHOHCH3 for 2-hydroxypropyl cyclodextrins
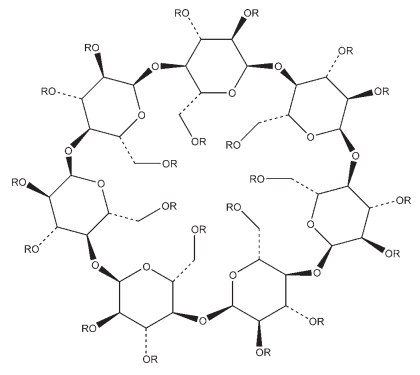
Cyclodextrins are crystalline, nonhygroscopic, cyclic oligosaccharides derived from starch. Among the most commonly used forms are a-, b-, and g-cyclodextrin, which have respectively 6, 7, and 8 glucose units; see Section 5. Substituted cyclodextrin derivatives are also available; see Section 17. Cyclodextrins are ‘bucketlike’ or ‘conelike’ toroid molecules, with a rigid structure and a central cavity, the size of which varies according to the cyclodextrin type; see Section 8. The internal surface of the cavity is hydrophobic and the outside of the torus is hydrophilic; this is due to the arrangement of hydroxyl groups within the molecule. This arrangement permits the cyclodextrin to accommodate a guest molecule within the cavity, forming an inclusion complex. Cyclodextrins may be used to form inclusion complexes with a variety of drug molecules, resulting primarily in improvements to dissolution and bioavailability owing to enhanced solubility and improved chemical and physical stability; see Section 18. Cyclodextrin inclusion complexes have also been used to mask the unpleasant taste of active materials and to convert a liquid substance into a solid material. b-Cyclodextrin is the most commonly used cyclodextrin, although it is the least soluble; see Section 10. It is the least expensive cyclodextrin; is commercially available from a number of sources; and is able to form inclusion complexes with a number of molecules of pharmaceutical interest. However, b-cyclodextrin is nephrotoxic and should not be used in parenteral formulations; see Section 14. b-Cyclodextrin is primarily used in tablet and capsule formulations. a-Cyclodextrin is used mainly in parenteral formulations. However, as it has the smallest cavity of the cyclodextrins it can form inclusion complexes with only relatively few, small-sized molecules. In contrast, g-cyclodextrin has the largest cavity and can be used to form inclusion complexes with large molecules; it has low toxicity and enhanced water solubility. In oral tablet formulations, b-cyclodextrin may be used in both wet-granulation and direct-compression processes. The physical properties of b-cyclodextrin vary depending on the manufacturer. However, b-cyclodextrin tends to possess poor flow properties and requires a lubricant, such as 0.1% w/w magnesium stearate, when it is directly compressed.(1,2) In parenteral formulations, cyclodextrins have been used to produce stable and soluble preparations of drugs that would otherwise have been formulated using a nonaqueous solvent. In eye drop formulations, cyclodextrins form water-soluble complexes with lipophilic drugs such as corticosteroids. They have been shown to increase the water solubility of the drug; to enhance drug absorption into the eye; to improve aqueous stability; and to reduce local irritation.(3) Cyclodextrins have also been used in the formulation of solutions,(4,5) suppositories,(6,7) and cosmetics.(8,9)
Cyclodextrins are cyclic oligosaccharides containing at least six D- (þ)-glucopyranose units attached by a(1!4) glucoside bonds. The three natural cyclodextrins, a, b, and g, differ in their ring size and solubility. They contain 6, 7, or 8 glucose units, respectively. Cyclodextrins occur as white, practically odorless, fine crystalline powders, having a slightly sweet taste. Some cyclodextrin derivatives occur as amorphous powders. See also Table I

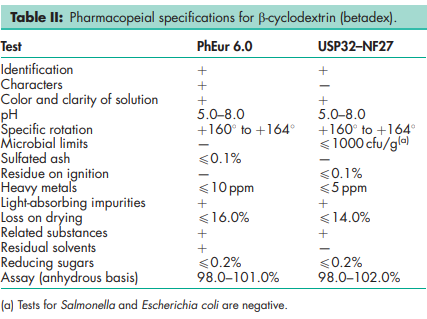
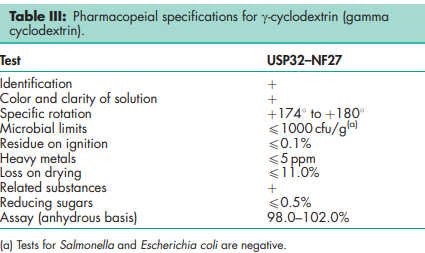
See Tables I, II, and III.
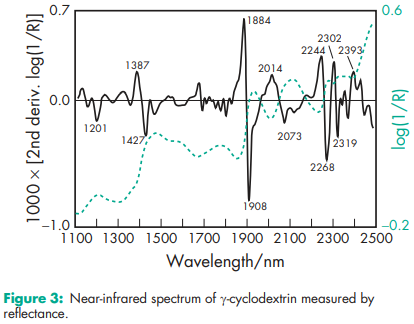
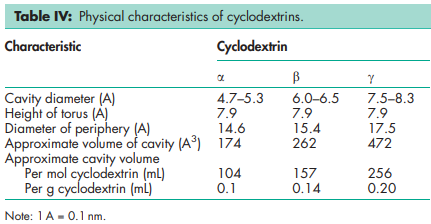
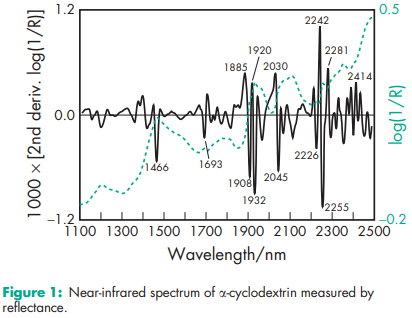
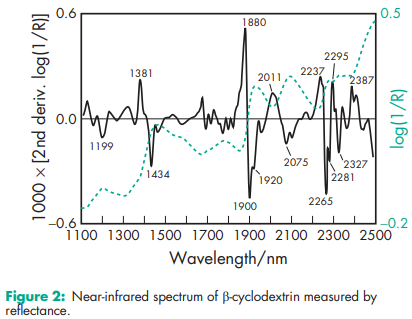
Compressibility 21.0–44.0% for b-cyclodextrin. Density (bulk) a-cyclodextrin: 0.526 g/cm3 ; b-cyclodextrin: 0.523 g/cm3 ; g-cyclodextrin: 0.568 g/cm3 . Density (tapped) a-cyclodextrin: 0.685 g/cm3 ; b-cyclodextrin: 0.754 g/cm3 ; g-cyclodextrin: 0.684 g/cm3 Density (true) a-cyclodextrin: 1.521 g/cm3 ; g-cyclodextrin: 1.471 g/cm3 . Melting point a-cyclodextrin: 250–2608C; b-cyclodextrin: 255–2658C; g-cyclodextrin: 240–2458C. Moisture content a-cyclodextrin: 10.2% w/w; b-cyclodextrin: 13.0–15.0% w/w; g-cyclodextrin: 8–18% w/w. NIR spectra see Figures 1, 2, and 3. Particle size distribution b-cyclodextrin: 7.0–45.0 mm Physical characteristics see Table IV. Solubility a-cyclodextrin: soluble 1 in 7 parts of water at 208C, 1 in 3 at 508C. b-cyclodextrin: soluble 1 in 200 parts of propylene glycol, 1 in 50 of water at 208C, 1 in 20 at 508C; practically insoluble in acetone, ethanol (95%), and methylene chloride. g-cyclodextrin: soluble 1 in 4.4 parts of water at 208C, 1 in 2 at 458C. Specific rotation a-cyclodextrin: [a]D 25 = þ150.58; b-cyclodextrin: [a]D 25 = þ162.08; g-cyclodextrin: [a]D 25 = þ177.48. Surface tension (at 258C) a-cyclodextrin: 71 mN/m (71 dynes/cm); b-cyclodextrin: 71 mN/m (71 dynes/cm); g-cyclodextrin: 71 mN/m (71 dynes/cm).
b-Cyclodextrin and other cyclodextrins are stable in the solid state if protected from high humidity. Cyclodextrins should be stored in a tightly sealed container, in a cool, dry place.
The activity of some antimicrobial preservatives in aqueous solution can be reduced in the presence of hydroxypropyl-b- cyclodextrin.(10–12)
Cyclodextrins are manufactured by the enzymatic degradation of starch using specialized bacteria. For example, b-cyclodextrin is produced by the action of the enzyme cyclodextrin glucosyltransferase upon starch or a starch hydrolysate. An organic solvent is used to direct the reaction that produces b-cyclodextrin, and to prevent the growth of microorganisms during the enzymatic reaction. The insoluble complex of b-cyclodextrin and organic solvent is separated from the noncyclic starch, and the organic solvent is removed in vacuo so that less than 1 ppm of solvent remains in the b-cyclodextrin. The b-cyclodextrin is then carbon treated and crystallized from water, dried, and collected.
Cyclodextrins are starch derivatives and are mainly used in oral and parenteral pharmaceutical formulations. They are also used in topical and ophthalmic formulations.(3) Cyclodextrins are also used in cosmetics and food products, and are generally regarded as essentially nontoxic and nonirritant materials. However, when administered parenterally, b-cyclodextrin is not metabolized but accumulates in the kidneys as insoluble cholesterol complexes, resulting in severe nephrotoxicity.(13) Cyclodextrin administered orally is metabolized by microflora in the colon, forming the metabolites maltodextrin, maltose, and glucose; these are themselves further metabolized before being finally excreted as carbon dioxide and water. Although a study published in 1957 suggested that orally administered cyclodextrins were highly toxic,(14) more recent animal toxicity studies in rats and dogs have shown this not to be the case, and cyclodextrins are now approved for use in food products and orally administered pharmaceuticals in a number of countries. Cyclodextrins are not irritant to the skin and eyes, or upon inhalation. There is also no evidence to suggest that cyclodextrins are mutagenic or teratogenic. a-Cyclodextrin LD50 (rat, IP): 1.0 g/kg(15) LD50 (rat, IV): 0.79 g/kg b-Cyclodextrin LD50 (mouse, IP): 0.33 g/kg(16) LD50 (mouse, SC): 0.41 g/kg LD50 (rat, IP): 0.36 g/kg LD50 (rat, IV): 1.0 g/kg LD50 (rat, oral): 18.8 g/kg LD50 (rat, SC): 3.7 g/kg g-Cyclodextrin LD50 (rat, IP): 4.6 g/kg(15) LD50 (rat ,IV): 4.0 g/kg LD50 (rat, oral): 8.0 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Cyclodextrins are fine organic powders and should be handled in a well-ventilated environment. Efforts should be made to limit the generation of dust, which can be explosive.
Included in the FDA Inactive Ingredients Database: a-cyclodextrin (injection preparations); b-cyclodextrin (oral tablets, topical gels); g-cyclodextrin (IV injections). Included in the Canadian List of Acceptable Non-medicinal Ingredients (stabilizing agent; solubilizing agent ); and in oral and rectal pharmaceutical formulations licensed in Europe, Japan, and the USA
Dimethyl-b-cyclodextrin; 2-hydroxyethyl-b-cyclodextrin; hydroxypropyl betadex; sulfobutylether b-cyclodextrin; trimethyl-b-cyclodextrin. Dimethyl-b-cyclodextrin Molecular weight 1331 Synonyms DM-b-CD. Appearance White crystalline powder. Cavity diameter 6 A˚ Melting point 295.0–300.08C Moisture content 41% w/w Solubility Soluble 1 in 135 parts of ethanol (95%), and 1 in 1.75 of water at 258C. Solubility decreases with increasing temperature. Surface tension 62 mN/m (62 dynes/cm) at 258C. Method of manufacture Dimethyl-b-cyclodextrin is prepared from b-cyclodextrin by the selective methylation of all C2 secondary hydroxyl groups and all C6 primary hydroxyl groups (C3 secondary hydroxyl groups remain unsubstituted). Comments Used in applications similar to those for b-cyclodextrin. 2-Hydroxyethyl-b-cyclodextrin CAS number [98513-20-3] Synonyms 2-HE-b-CD. Appearance White crystalline powder. Density (bulk) 0.681 g/cm3 Density (tapped) 0.916 g/cm3 Density (true) 1.378 g/cm3 Solubility Greater than 1 in 2 parts of water at 258C. Surface tension 68.0–71.0 mN/m (68–71 dynes/cm) at 258C. Comments Used in applications similar to those for b-cyclodextrin. The degree of substitution of hydroxyethyl groups can vary.(17) Trimethyl-b-cyclodextrin Molecular weight 1429 Synonyms TM-b-CD. Appearance White crystalline powder. Cavity diameter 4.0–7.0 A˚ Melting point 1578C Moisture content 41% w/w Solubility Soluble 1 in 3.2 parts of water at 258C. Solubility decreases with increasing temperature. Surface tension 56 mN/m (56 dynes/cm) at 258C. Method of manufacture Trimethyl-b-cyclodextrin is prepared from b-cyclodextrin by the complete methylation of all C2 and C3 secondary hydroxyl groups along with all C6 primary hydroxyl groups. Comments Used in applications similar to those for b-cyclodextrin
In addition to their use in pharmaceutical formulations, cyclodextrins have also been investigated for use in various industrial applications. Analytically, cyclodextrin polymers are used in chromatographic separations, particularly of chiral materials. b-Cyclodextrin derivatives are more water-soluble than bcyclodextrin, and studies have shown that they have greater solubilizing action with some drugs such as ibuproxam, a poorly water-soluble anti-inflammatory agent.(18,19) The EINECS number for cyclodextrin is 231-493-2. The PubChem Compound ID (CID) for cyclodextrins includes 444913 (a-cyclodextrin), 24238 (b-cyclodextrin), and 86575 (g-cyclodextrin).