

Name: Cyclomethicone
CAS No: Cyclopolydimethylsiloxane [69430-24-6]
USP-NF: Cyclomethicone
Dimethylcyclopolysiloxane; Dow Corning 245 Fluid; Dow Corning 246 Fluid; Dow Corning 345 Fluid.
Cyclopolydimethylsiloxane [69430-24-6]
The USP32–NF27 describes cyclomethicone as a fully methylated cyclic siloxane containing repeating units of the formula [– (CH3)2SiO–]n in which n is 4, 5, or 6, or a mixture of them
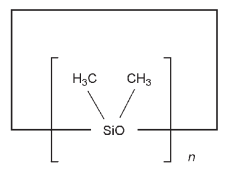
Cyclomethicone is mainly used in topical pharmaceutical and cosmetic formulations such as water-in-oil creams.(1–3) Cyclomethicone has been used in cosmetic formulations, at concentrations of 0.1–50%, since the late 1970s and is now the most widely used silicone in the cosmetics industry. Its high volatility, and mild solvent properties, make it ideal for use in topical formulations because its low heat of vaporization means that when applied to skin it has a ‘dry’ feel. See also Dimethicone.
Cyclomethicone occurs as a clear, colorless and tasteless volatile liquid.
See Table I.
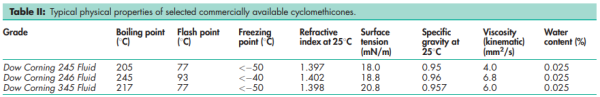

Solubility Soluble in ethanol (95%), isopropyl myristate, isopropyl palmitate, mineral oil, and petrolatum at 808C; practially insoluble in glycerin, propylene glycol, and water. See also Table II.
Cyclomethicone should be stored in an airtight container in a cool, dry, place.
Cyclomethicone is manufactured by the distillation of crude polydimethylsiloxanes
Cyclomethicone is generally regarded as a relatively nontoxic and nonirritant material. Although it has been used in oral pharmaceutical applications, cyclomethicone is mainly used in topical pharmaceutical formulations. It is also widely used in cosmetics.(4) Studies of the animal and human toxicology of cyclomethicone suggest that it is nonirritant and not absorbed through the skin. Only small amounts are absorbed orally; an acute oral dose in rats produced no deaths.(5,6) See also Dimethicone.
Observe normal precautions appropriate to the circumstances and quantity of material handled.
Included in the FDA Inactive Ingredients Database (oral powder for reconstitution, topical lotion, topical cream, topical emulsion). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Dimethicone; simethicone