

Name: Isopropyl Alcohol
CAS No: Propan-2-ol [67-63-0]
BP: Isopropyl Alcohol JP: Isopropanol PhEur: Isopropyl Alcohol USP: Isopropyl Alcohol
Alcohol isopropylicus; dimethyl carbinol; IPA; isopropanol; petrohol; 2-propanol; sec-propyl alcohol; rubbing alcohol.
Propan-2-ol [67-63-0]
C3H8O 60.1
Isopropyl alcohol (propan-2-ol) is used in cosmetics and pharmaceutical formulations, primarily as a solvent in topical formulations.(1) It is not recommended for oral use owing to its toxicity; see Section 14. Although it is used in lotions, the marked degreasing properties of isopropyl alcohol may limit its usefulness in preparations used repeatedly. Isopropyl alcohol is also used as a solvent both for tablet film-coating and for tablet granulation,(2) where the isopropyl alcohol is subsequently removed by evaporation. It has also been shown to significantly increase the skin permeability of nimesulide from carbomer 934.(3) Isopropyl alcohol has some antimicrobial activity (see Section 10) and a 70% v/v aqueous solution is used as a topical disinfectant. Therapeutically, isopropyl alcohol has been investigated for the treatment of postoperative nausea or vomiting.(4)
Isopropyl alcohol is a clear, colorless, mobile, volatile, flammable liquid with a characteristic, spirituous odor resembling that of a mixture of ethanol and acetone; it has a slightly bitter taste.
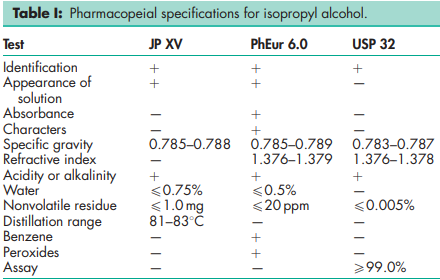
See Table I.
Antimicrobial activity Isopropyl alcohol is bactericidal; at concentrations greater than 70% v/v it is a more effective antibacterial preservative than ethanol (95%). The bactericidal effect of aqueous solutions increases steadily as the concentration approaches 100% v/v. Isopropyl alcohol is ineffective against bacterial spores. Autoignition temperature 4258C Boiling point 82.48C Dielectric constant D20 = 18.62 Explosive limits 2.5–12.0% v/v in air Flammability Flammable. Flash point 11.78C (closed cup); 138C (open cup). The water azeotrope has a flash point of 168C. Freezing point 89.58C Melting point 88.58C Moisture content 0.1–13% w/w for commercial grades (13% w/w corresponds to the water azeotrope). Refractive index nD 20 = 1.3776; nD 25 = 1.3749. Solubility Miscible with benzene, chloroform, ethanol (95%), ether, glycerin, and water. Soluble in acetone; insoluble in salt solutions. Forms an azeotrope with water, containing 87.4% w/w isopropyl alcohol (boiling point 80.378C). Specific gravity 0.786 Vapor density (relative) 2.07 (air = 1) Vapor pressure 133.3 Pa (1 mmHg) at 26.18C; 4.32 kPa (32.4 mmHg) at 208C; 5.33 kPa (40 mmHg) at 23.88C; 13.33 kPa (100 mmHg) at 39.58C. Viscosity (dynamic) 2.43 mPa s (2.43 cP) at 208C
Isopropyl alcohol should be stored in an airtight container in a cool, dry place.
Incompatible with oxidizing agents such as hydrogen peroxide and nitric acid, which cause decomposition. Isopropyl alcohol may be salted out from aqueous mixtures by the addition of sodium chloride, sodium sulfate, and other salts, or by the addition of sodium hydroxide
Isopropyl alcohol may be prepared from propylene; by the catalytic reduction of acetone, or by fermentation of certain carbohydrates
Isopropyl alcohol is widely used in cosmetics and topical pharmaceutical formulations. It is readily absorbed from the gastrointestinal tract and may be slowly absorbed through intact skin. Prolonged direct exposure of isopropyl alcohol to the skin may result in cardiac and neurological deficits.(5) In neonates, isopropyl alcohol has been reported to cause chemical burns following topical application.(6,7) Isopropyl alcohol is metabolized more slowly than ethanol, primarily to acetone. Metabolites and unchanged isopropyl alcohol are mainly excreted in the urine. Isopropyl alcohol is about twice as toxic as ethanol and should therefore not be administered orally; isopropyl alcohol also has an unpleasant taste. Symptoms of isopropyl alcohol toxicity are similar to those for ethanol except that isopropyl alcohol has no initial euphoric action, and gastritis and vomiting are more prominent; see Alcohol. Delta osmolality may be useful as rapid screen test to identify patients at risk of complications from ingestion of isopropyl alcohol.(8) The lethal oral dose is estimated to be about 120–250 mL although toxic symptoms may be produced by 20 mL. Adverse effects following parenteral administration of up to 20 mL of isopropyl alcohol diluted with water have included only a sensation of heat and a slight lowering of blood pressure. However, isopropyl alcohol is not commonly used in parenteral products. Although inhalation can cause irritation and coma, the inhalation of isopropyl alcohol has been investigated in therapeutic applications.(3) Isopropyl alcohol is most frequently used in topical pharmaceutical formulations where it may act as a local irritant.(9) When applied to the eye it can cause corneal burns and eye damage. LD50 (dog, oral): 4.80 g/kg(9) LD50 (mouse, oral): 3.6 g/kg LD50 (mouse, IP): 4.48 g/kg LD50 (mouse, IV): 1.51 g/kg LD50 (rabbit, oral): 6.41 g/kg LD50 (rabbit, skin): 12.8 g/kg LD50 (rat, IP): 2.74 g/kg LD50 (rat, IV): 1.09 g/kg LD50 (rat, oral): 5.05 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Isopropyl alcohol may be irritant to the skin, eyes, and mucous membranes upon inhalation. Eye protection and gloves are recommended. Isopropyl alcohol should be handled in a well-ventilated environment. In the UK, the longterm (8-hour TWA) workplace exposure limit for isopropyl alcohol is 999 mg/m3 (400 ppm); the short-term (15-minute) workplace exposure limit is 1250 mg/m3 (500 ppm).(10) OSHA standards state that IPA 8-hour time weighted average airborne level in the workplace cannot exceed 400 ppm. Isopropyl alcohol is flammable and produces toxic fumes on combustion.
Included in the FDA Inactive Ingredients Database (oral capsules, tablets, and topical preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Propan-1-ol. Propan-1-ol Empirical formula C3H8O Molecular weight 60.1 CAS number [71-23-8] Synonyms Propanol; n-propanol; propyl alcohol; propylic alcohol. Autoignition temperature 5408C Boiling point 97.28C Dielectric constant D 25 = 22.20 Explosive limits 2.15–13.15% v/v in air Flash point 158C (closed cup) Melting point –1278C Refractive index n D 20 = 1.3862 Solubility Miscible with ethanol (95%), ether, and water. Specific gravity 0.8053 at 208C Viscosity (dynamic) 2.3 mPa s (2.3 cP) at 208C Comments Propan-1-ol is more toxic than isopropyl alcohol. In the UK, the long-term (8-hour TWA) exposure limit for propan1-ol is 500 mg/m3 (200 ppm); the short-term (15-minute) exposure limit is 625 mg/m3 (250 ppm).(10)
A specification for isopropyl alcohol is contained in the Food Chemicals Codex (FCC).(11) The EINECS number for isopropyl alcohol is 200-661-7. The PubChem Compound ID (CID) for isopropyl alcohol is 3776.