

Name: Kaolin
CAS No: Hydrated aluminum silicate [1332-58-7]
BP: Heavy Kaolin JP: Kaolin PhEur: Kaolin, Heavy USP: Kaolin Note that the PhEur 6.3 contains a monograph on heavy kaolin. The BP 2009, in addition to the monograph for heavy kaolin, also contains monographs for light kaolin (natural) and light kaolin
Argilla; bolus alba; China clay; E559; kaolinite; kaolinum ponderosum; Lion; porcelain clay; Sim 90; weisserton; white bole.
Hydrated aluminum silicate [1332-58-7]
Al2H4O9Si2 258.16 The USP 32 describes kaolin as a native hydrated aluminum silicate, powdered and freed from gritty particles by elutriation. The BP 2009 similarly describes light kaolin but additionally states that it contains a suitable dispersing agent. Light kaolin (natural) BP contains no dispersing agent. Heavy kaolin is described in the BP 2009 and PhEur 6.3 as a purified, natural hydrated aluminum silicate of variable composition. The JP XV describes kaolin as a native hydrous aluminum silicate.
See Section 4.
Kaolin is a naturally occurring mineral used in oral and topical pharmaceutical formulations. In oral medicines, kaolin has been used as a diluent in tablet and capsule formulations; it has also been used as a suspending vehicle. In topical preparations, sterilized kaolin has been used in poultices and as a dusting powder. Therapeutically, kaolin has been used in oral antidiarrheal preparations.(1,2
Kaolin occurs as a white to grayish-white colored, unctuous powder free from gritty particles. It has a characteristic earthy or claylike taste, and when moistened with water it becomes darker in color and develops a claylike odor.


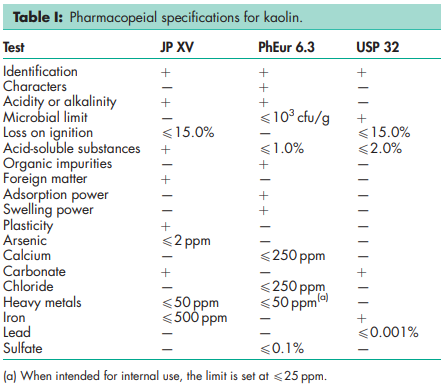
See Table I
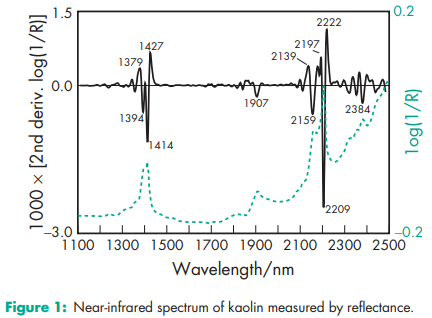
Acidity/alkalinity pH = 4.0–7.5 for a 20% w/v aqueous slurry Hardness (Mohs) 2.0, very low Hygroscopicity At relative humidities between about 15–65%, the equilibrium moisture content at 258C is about 1% w/w, but at relative humidities above about 75%, kaolin absorbs small amounts of moisture. NIR spectra see Figure 1. Particle size distribution Median size = 0.6–0.8 mm Refractive index 1.56 Solubility Practically insoluble in diethyl ether, ethanol (95%), water, other organic solvents, cold dilute acids, and solutions of alkali hydroxides. Specific gravity 2.6 Viscosity (dynamic) 300 mPa s (300 cP) for a 70% w/v aqueous suspension Whiteness 85–90% of the brightness of MgO
Kaolin is a stable material. Since it is a naturally occurring material, kaolin is commonly contaminated with microorganisms such as Bacillus anthracis, Clostridium tetani, and Clostridium welchii. However, kaolin may be sterilized by heating at a temperature greater than 1608C for not less than 1 hour. When moistened with water, kaolin darkens and becomes plastic. Kaolin should be stored in a well-closed container in a cool, dry place.
The adsorbent properties of kaolin may influence the absorption of other orally administered drugs. Drugs reportedly affected by kaolin include amoxicillin;(3) ampicillin;(3) cimetidine;(4) digoxin;(5) lincomycin; phenytoin;(6) and tetracycline. Warfarin absorption by rat intestine in vitro was reported not to be affected by kaolin.(7) With clindamycin, the rate (but not the amount) of absorption was affected by kaolin.(8)
Kaolin is a hydrated aluminum silicate obtained by mining naturally occurring mineral deposits. Large deposits are found in Georgia, USA and in Cornwall, England. Mined kaolin is powdered and freed of coarse, gritty particles either by elutriation or by screening. Impurities such as ferric oxide, calcium carbonate, and magnesium carbonate are removed with an electromagnet and by treatment with hydrochloric acid and/or sulfuric acids.
Kaolin is used in oral and topical pharmaceutical formulations and is generally regarded as an essentially nontoxic and nonirritant material. Oral doses of about 2–6 g of kaolin every 4 hours have been administered in the treatment of diarrhea.(1,2)
Observe normal precautions appropriate to the circumstances and quantity of material handled. The chronic inhalation of kaolin dust can cause diseases of the lung (silicosis or kaolinosis).(9) Eye protection and a dust mask are recommended. In the UK, the longterm (8-hour TWA) workplace exposure limit for kaolin respirable dust is 2 mg/m3 . (10)
Accepted in Europe as a food additive in certain applications. Included in the FDA Inactive Ingredients Database (oral capsules, powders, syrups, and tablets; topical preparations). Included in nonparenteral medicines licensed in the UK
Bentonite; magnesium aluminum silicate.
Kaolin is considered in most countries to be an archaic diluent. The name kaolinite was historically used to describe the processed mineral, while the name kaolin was used for the unprocessed clay. However, the two names have effectively become synonymous and kaolin is now generally the only name used. A specification for kaolin is contained in the Food Chemicals Codex (FCC).(11) The EINECS number for kaolin is 310-127-6.