

Name: Lactose, Anhydrous
CAS No: O-b-D-Galactopyranosyl-(1!4), b-D-glucopyranose [63-42-3]
BP: Anhydrous Lactose JP: Anhydrous Lactose PhEur: Lactose, Anhydrous USP-NF: Anhydrous Lactose
Anhydrous 60M; Anhydrous Direct Tableting (DT); Anhydrous DT High Velocity; Anhydrous Impalpable; Lactopress Anhydrous; Lactopress Anhydrous 250; lactosum anhydricum; lattosio; milk sugar; SuperTab 21AN; SuperTab 22AN; saccharum lactis
O-b-D-Galactopyranosyl-(1!4), b-D-glucopyranose [63-42-3]
C12H22O11 342.30
The PhEur 6.5 and USP32–NF27 describe anhydrous lactose as Ob-D-galactopyranosyl-(1!4)-b-D-glucopyranose; or a mixture of O-b-D-galactopyranosyl-(1!4)-a-D-glucopyranose and O-b-Dgalactopyranosyl-(1!4)-b-D-glucopyranose. The JP XV describes anhydrous lactose as b-lactose or a mixture of b-lactose and alactose, and defines these as per the PhEur and USP–NF
Anhydrous lactose is widely used in direct compression tableting applications, and as a tablet and capsule filler and binder. Anhydrous lactose can be used with moisture-sensitive drugs due to its low moisture content. It may also be used in intravenous injections See also Lactose, Inhalation; Lactose, Monohydrate; Lactose, Spray-Dried
Anhydrous lactose occurs as white to off-white crystalline particles or powder. Several different brands of anhydrous lactose are commercially available which contain anhydrous b-lactose and anhydrous a-lactose. Anhydrous lactose typically contains 70–80% anhydrous b-lactose and 20–30% anhydrous a-lactose.
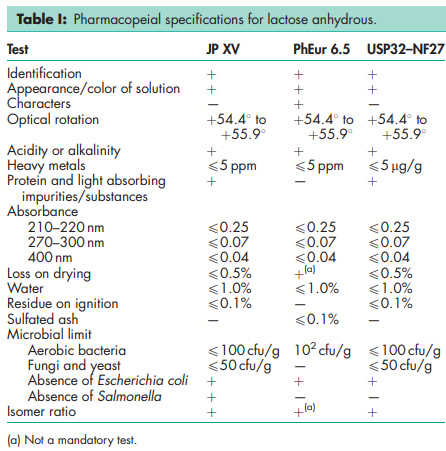
See Table I.
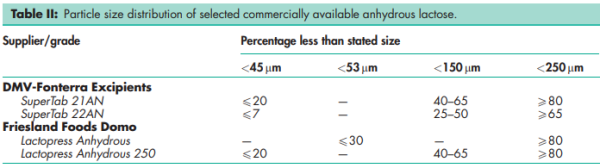
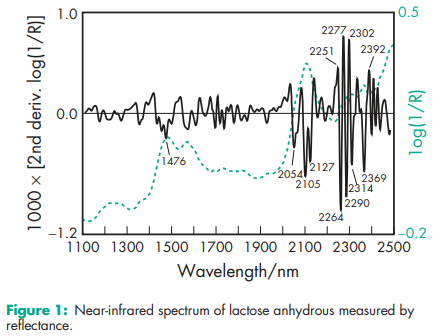
Brittle fracture index 0.0362 Bonding index 0.0049 (at compression pressure 177.8 MPa)(1) Density (true) 1.589 g/cm3 for anhydrous b-lactose Density (bulk) 0.71 g/cm3 for SuperTab 21AN; 0.66 g/cm3 for Super-Tab 22AN. Density (tapped) 0.88 g/cm3 for SuperTab 21AN; 0.78 g/cm3 for Super-Tab 22AN. Melting point 223.08C for anhydrous a-lactose; 252.28C for anhydrous b-lactose; 232.08C (typical) for commercial anhydrous lactose. NIR spectra see Figure 1. Particle size distribution see Table II. Permanent deformation pressure 521.0MPa (at compression pressure 177.8 MPa)(1) Reduced modulus of elasticity 5315 (at compression pressure 177.8 MPa)(1) Solubility Soluble in water; sparingly soluble in ethanol (95%) and ether; 40 g/100 mL at 258C for typical Sheffield Pharma Ingredients products. Specific rotation [a]D 25 = 54.48 to 55.98 Tensile strength 2.577 MPa (at compression pressure 177.8 MPa)(1) Methods for characterizing the mechanical properties of compacts of pharmaceutical ingredients are specified in the Handbook of Pharmaceutical Excipients, 3rd edn.(1)


Mold growth may occur under humid conditions (80% RH and above). Lactose may develop a brown coloration on storage, the reaction being accelerated by warm, damp conditions; see Section 12. At 808C and 80% RH, tablets containing anhydrous lactose have been shown to expand 1.2 times after one day.(2) Lactose anhydrous should be stored in a well-closed container in a cool, dry place.
Lactose anhydrous is incompatible with strong oxidizers. When mixtures containing a hydrophobic leukotriene antagonist and anhydrous lactose or lactose monohydrate were stored for six weeks at 408C and 75% RH, the mixture containing anhydrous lactose showed greater moisture uptake and drug degradation.(3) Studies have also shown that in blends of roxifiban acetate (DMP-754) and lactose anhydrous, the presence of lactose anhydrous accelerated the hydrolysis of the ester and amidine groups.(4) Lactose anhydrous is a reducing sugar with the potential to interact with primary(5) and secondary amines(6) (Maillard reaction) when stored under conditions of high humidity for extended periods. See Lactose, Monohydrate.
There are two anhydrous forms of lactose: a-lactose and b-lactose. The temperature of crystallization influences the ratio of a- and blactose. The anhydrous forms that are commercially available may exhibit hygroscopicity at high relative humidities. Anhydrous lactose is produced by roller drying a solution of lactose above 93.58C. The resulting product is then milled and sieved. Two anhydrous a-lactoses can be prepared using special drying techniques: one is unstable and hygroscopic; the other exhibits good compaction properties.(7) However, these materials are not commercially available.
Lactose is widely used in pharmaceutical formulations as a diluent and filler-binder in oral capsule and tablet formulations. It may also be used in intravenous injections. Adverse reactions to lactose are largely due to lactose intolerance, which occurs in individuals with a deficiency of the intestinal enzyme lactase, and is associated with oral ingestion of amounts well over those found in solid dosage forms
Observe normal precautions appropriate to the circumstances and quantity of materials handled. Excessive generation of dust, or inhalation of dust, should be avoided.
GRAS listed. Included in the FDA Inactive Ingredients Database (IM, IV: powder for injection solution; IV and sublingual preparations; oral: capsules and tablets; powder for inhalation; vaginal). Included in nonparenteral and parenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredient
Lactose, inhalation; lactose, monohydrate; lactose, spray-dried
Lactose anhydrous is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.5, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. Lactose anhydrous has been used experimentally in hydrophilic matrix tablet formulations(8) and evaluated for dry powder inhalation applications.(9,10) Partial hydration of anhydrous lactose increases the specific surface area and reduces the flow properties of powders but has no effect on compactibility.(11) A specification for lactose is included in the Food Chemicals Codex (FCC(12)); see Lactose, Monohydrate. The EINECS number for lactose anhydrous is 200-559-2. The PubChem Compound ID (CID) for lactose anhydrous includes 6134 and 84571.