

Name: Lanolin, Hydrous
CAS No: Hydrous lanolin [8020-84-6
BP: Hydrous Wool Fat JP: Hydrous Lanolin PhEur: Wool Fat, Hydrous
Adeps lanae cum aqua; Lipolan.
Hydrous lanolin [8020-84-6
The JP XV describes hydrous lanolin as a mixture of lanolin and 25–30% w/w purified water. The PhEur 6.0 describes hydrous lanolin as a mixture of lanolin and 25% w/w purified water; see also Section 18. The PhEur 6.0 additionally permits the inclusion of up to 150 ppm of butylated hydroxytoluene as an antioxidant
See Section 4.
Hydrous lanolin is widely used in topical pharmaceutical formulations and cosmetics in applications similar to those for lanolin. Hydrous lanolin is commonly used in the preparation of waterin-oil creams and ointments. More water may be incorporated into hydrous lanolin than into lanolin.
Hydrous lanolin is a pale yellow-colored, unctuous substance with a faint characteristic odor. When melted by heating on a water bath hydrous lanolin separates into a clear oily layer and a clear water layer.
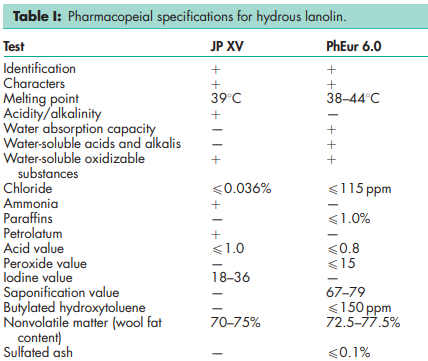
See Table I.
Solubility Practically insoluble in chloroform, ether, and water. Only the fat component of hydrous lanolin is soluble in organic solvents
Hydrous lanolin should be stored in a well-filled, well-closed container protected from light, in a cool, dry place. Normal storage life is 2 years.
See Lanolin
Lanolin is melted, and sufficient purified water is gradually added with constant stirring.
Hydrous lanolin is used in cosmetics and a number of topical pharmaceutical formulations and is generally regarded as a nontoxic and nonirritant material, although it has been associated with hypersensitivity reactions. See Lanolin for further information
Observe normal precautions appropriate to the circumstances and quantity of material handled.
Included in the FDA Inactive Ingredients Database (ophthalmic, topical, transdermal, and vaginal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Cholesterol; lanolin; lanolin alcohols.
Lanolin (the anhydrous material) may be confused in some instances with hydrous lanolin since the USP formerly contained monographs for ‘lanolin’ and ‘anhydrous lanolin’ in which the name ‘lanolin’ referred to the material containing 25–30% w/w of purified water.