

Name: Macrogol 15 Hydroxystearate
CAS No: 2-Hydroxyethyl, 12-hydroxyoctadecanoate [70142-34-6]
BP: Macrogol 15 Hydroxystearate PhEur: Macrogol 15 Hydroxystearate
12-Hydroxyoctadecanoic acid polymer with a-hydro-o-hydroxypoly(oxy-1,2-ethanediyl); 12-hydroxystearic acid polyethylene glycol copolymer; macrogoli 15 hydroxystearas; polyethylene glycol15-hydroxystearate; polyethylene glycol 660 12-hydroxystearate; Solutol HS 15.
2-Hydroxyethyl, 12-hydroxyoctadecanoate [70142-34-6]
The PhEur 6.0 describes macrogol 15 hydroxystearate as a mixture of mainly monoesters and diesters of 12-hydroxystearic acid and macrogols obtained by the ethoxylation of 12-hydroxystearic acid. The number of moles of ethylene oxide reacted per mole of 12- hydroxystearic acid is 15 (nominal value). It contains about 30% free macrogols. C20H40O4 344.53
See Section 4.
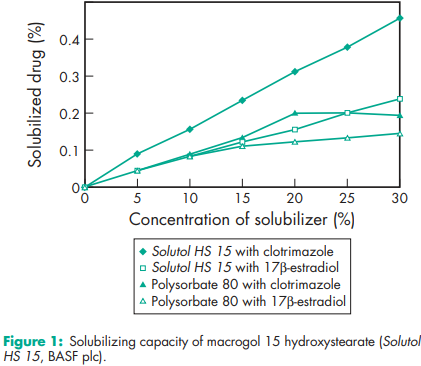
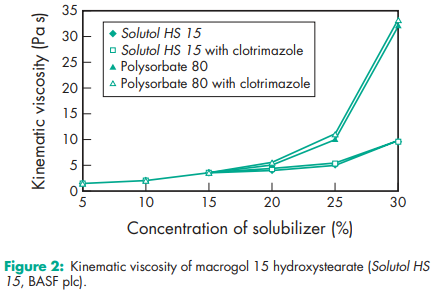
Macrogol 15 hydroxystearate is frequently used in preclinical testing of drugs, mainly for IV and other parenteral applications.(1–4) The solubilizing capacity for some tested drugs (clotrimazole, carbamazepine, 17b-estradiol, sulfathiazole, and piroxicam) increases almost linearly with increasing concentration of solubilizing agent; see Figure 1. This is due to the formation of spherical micelles even at high concentrations of macrogol 15 hydroxystearate. Similarly, tests have revealed that viscosity increases with increasing amount of solubilizer, but the amount of solubilized drugs does not have any additional influence on the kinematic viscosity; see Figure 2. Lipid nanocapsules comprising macrogol 15 hydroxystearate and soybean phosphatidylcholine containing 3% docetaxel have been successfully prepared by a solvent-free inversion process. Macrogol 15 hydroxystearate has been used in the manufacture of aqueous parenteral preparations with vitamin A, D, E and K, and a number of other lipophilic pharmaceutical active agents, such as propanidid, miconazole, alfadolone, and alfaxalone. It is very efficient at solubilizing substances like fat-soluble vitamins and active ingredients of hydrophobic nature. It is also an excellent solubilizer for parenteral use, at a concentration of 20%, and the water solubility of different drugs may be enhanced by a factor of 10–100, depending on the structure of the drug molecule.
Macrogol 15 hydroxystearate is a yellowish-white, almost odorless waxy mass or paste at room temperature, which becomes liquid at approximately 308C.
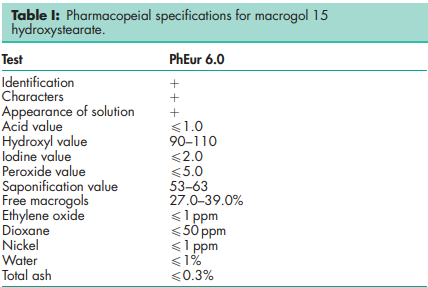
See Table I.
Acidity/alkalinity pH = 6–7 (10% w/v aqueous solution at 208C) Critical micelle concentration 0.005–0.02% Density 1.03 g/cm3 Flash point 2728C HLB value 14–16 Ignition temperature 3608C Solidification temperature 25–308C Solubility Soluble in organic solvents such as ethanol (95%), propan-2-ol, and very soluble in water to form clear solutions. The solubility in water decreases with increasing temperature. It is insoluble in liquid paraffin. Viscosity (dynamic) 12 mPa s (12 cP) for a 30% w/v aqueous solution at 258C; 73 mPa s (73 cP) for a 30% w/v aqueous solution at 608C.
Macrogol 15 hydroxystearate has a high chemical stability. The prolonged action of heat may induce physical separation into a liquid and a solid phase after cooling, which can be reversed by subsequent homogenization. Macrogol 15 hydroxystearate is stable for at least 24 months if stored in unopened airtight containers at room temperature (maximum 258C). Aqueous solutions of macrogol 15 hydroxystearate can be heat-sterilized (1218C, 0.21 MPa). The pH may drop slightly during heating, which should be taken into account. Separation into phases may also occur, but agitating the hot solution can reverse this. Aqueous solutions can be stabilized with the standard preservatives used in pharmaceuticals. Macrogol 15 hydroxystearate should be stored in tightly sealed containers in a dry place.
Macrogol 15 hydroxystearate is produced by reacting 15 moles of ethylene oxide with 1 mole of 12-hydroxystearic acid.
Macrogol 15 hydroxystearate is used in parenteral pharmaceutical preparations in concentrations up to 50% to solubilize diclofenac, propanidid, and vitamin K1. It has also been used in preclinical formulations in preparing supersaturated injectable formulations of water-insoluble molecules. It is generally regarded as a relatively nontoxic and nonirritant excipient. Macrogol 15 hydroxystearate is reported not to be mutagenic in bacteria, mammalian cell cultures and mammals. LD50 (dog, IV): >3.10 g/kg(5) LD50 (mouse, IP): >0.0085 g/kg LD50 (mouse, IV): >3.16 g/kg LD50 (rabbit, IV): 1.0–1.4 g/kg LD50 (rat, oral): >20 g/kg LD50 (rat, IV): 1.0–1.47 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled.
Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Polyethylene glycol
Macrogol 15 hydroxystearate is not restricted solely to parenteral use, but is also suitable for oral applications. Macrogol 15 hydroxystearate has been investigated as a coemulsifier in the preparation of parenteral o/w emulsions(2) and microemulsions.(6–8) It has also been investigated as a weak inhibitor of cytochrome P450 3A activity on the metabolism of colchicine and midazolum.(9–11) Oral bioavailability of the highly lipophilic and poorly watersoluble immunosuppressive agent, ciclosporin A, showed twofold higher bioavailability with a macrogol 15 hydroxystearate-based formulation compared to a microsuspension.(12) It has also been studied along with microcrystalline cellulose to prepare selfemulsifying pellets using an extrusion/spheronization technique to increase the bioavailability of lipophilic drugs.(13) Macrogol 15 hydroxystearate has been incorporated as a solubility-increasing additive in rectal suppository dosage form to study the increase in bioavailability of poorly water-soluble drugs.(14) Macrogol 15 hydroxystearate has been investigated as a therapeutic agent in the preparation of lipid nanoparticles of an anticancer drug,(15) and has also been shown to be effective for reversing multidrug resistance, with low toxicity in vivo. (16) The PubChem Compound ID (CID) for Solutol HS 15 is 124898.