

Name: Magnesium Stearate
CAS No: Octadecanoic acid magnesium salt [557-04-0]
BP: Magnesium Stearate JP: Magnesium Stearate PhEur: Magnesium Stearate USP-NF: Magnesium Stearate
Dibasic magnesium stearate; magnesium distearate; magnesii stearas; magnesium octadecanoate; octadecanoic acid, magnesium salt; stearic acid, magnesium salt; Synpro 90
Octadecanoic acid magnesium salt [557-04-0]
C36H70MgO4 591.24 The USP32–NF27 describes magnesium stearate as a compound of magnesium with a mixture of solid organic acids that consists chiefly of variable proportions of magnesium stearate and magnesium palmitate (C32H62MgO4). The PhEur 6.5 describes magnesium stearate as a mixture of solid organic acids consisting mainly of variable proportions of magnesium stearate and magnesium palmitate obtained from sources of vegetable or animal origin.
[CH3(CH2)16COO]2Mg
Magnesium stearate is widely used in cosmetics, foods, and pharmaceutical formulations. It is primarily used as a lubricant in capsule and tablet manufacture at concentrations between 0.25% and 5.0% w/w. It is also used in barrier creams.
Magnesium stearate is a very fine, light white, precipitated or milled, impalpable powder of low bulk density, having a faint odor of stearic acid and a characteristic taste. The powder is greasy to the touch and readily adheres to the skin.
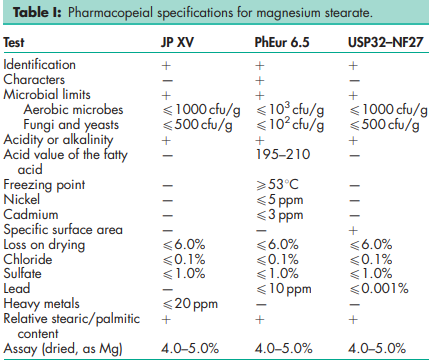
See Table I.
Crystalline forms High-purity magnesium stearate has been isolated as a trihydrate, a dihydrate, and an anhydrate. Density (bulk) 0.159 g/cm3 Density (tapped) 0.286 g/cm3 Density (true) 1.092 g/cm3 Flash point 2508C Flowability Poorly flowing, cohesive powder. Melting range 117–1508C (commercial samples) 126–1308C (high purity magnesium stearate). NIR spectra see Figure 1. Solubility Practically insoluble in ethanol, ethanol (95%), ether and water; slightly soluble in warm benzene and warm ethanol (95%). Specific surface area 1.6–14.8 m2 /g


Magnesium stearate is stable and should be stored in a well-closed container in a cool, dry place.
Incompatible with strong acids, alkalis, and iron salts. Avoid mixing with strong oxidizing materials. Magnesium stearate cannot be used in products containing aspirin, some vitamins, and most alkaloidal salts.
Magnesium stearate is prepared either by the interaction of aqueous solutions of magnesium chloride with sodium stearate or by the interaction of magnesium oxide, hydroxide, or carbonate with stearic acid at elevated temperatures.
Magnesium stearate is widely used as a pharmaceutical excipient and is generally regarded as being nontoxic following oral administration. However, oral consumption of large quantities may produce a laxative effect or mucosal irritation. No toxicity information is available relating to normal routes of occupational exposure. Limits for heavy metals in magnesium stearate have been evaluated in terms of magnesium stearate worstcase daily intake and heavy metal composition.(1) Toxicity assessments of magnesium stearate in rats have indicated that it is not irritating to the skin, and is nontoxic when administered orally or inhaled.(2,3) Magnesium stearate has not been shown to be carcinogenic when implanted into the bladder of mice.(4) LD50 (rat, inhalation): >2 mg/L(2) LD50 (rat, oral): >10 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended. Excessive inhalation of magnesium stearate dust may cause upper respiratory tract discomfort, coughing, and choking. Magnesium stearate should be handled in a well-ventilated environment; a respirator is recommended. In the USA, the OSHA limit is 10 mg/m3 TWA for magnesium stearate.
GRAS listed. Accepted as a food additive in the USA and UK. Included in the FDA Inactive Ingredients Database (oral capsules, powders, and tablets; buccal and vaginal tablets; topical prepara-tions; intravitreal implants and injections). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients. Listed on the US TSCA inventory.
Calcium stearate; magnesium aluminum silicate; stearic acid; zinc stearate.
Magnesium stearate is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. Magnesium stearate is hydrophobic and may retard the dissolution of a drug from a solid dosage form; the lowest possible concentration is therefore used in such formulations.(5–10) Capsule dissolution is also sensitive to both the amount of magnesium stearate in the formulation and the mixing time; higher levels of magnesium stearate and long mixing times can result in the formation of hydrophobic powder beds that do not disperse after the capsule shell dissolves.(11,12) An increase in the coefficient of variation of mixing and a decrease in the dissolution rate have been observed following blending of magnesium stearate with a tablet granulation. Tablet dissolution rate and crushing strength decreased as the time of blending increased; and magnesium stearate may also increase tablet friability. Blending times with magnesium stearate should therefore be carefully controlled.(13–29) A variety of online analytical techniques have been investigated to monitor magnesium stearate in powder blends and tablets.(30–32) Inverse gas chromatography has been used to examine the surface coverage of magnesium stearate on powder blends.(33) Magnesium stearate also affects the flow properties of blends.(34) The existence of various crystalline forms of magnesium stearate has been established.(35–39) A trihydrate, a dihydrate, and an anhydrate have been isolated,(5,37,38,40) and an amorphous form has been observed.(41) While the hydrated forms are stable in the presence of moisture, the anhydrous form adsorbs moisture at relative humidity up to 50%, and at higher humidities rehydrates to form the trihydrate. The anhydrate can be formed by drying either of the hydrates at 1058C.(38) It has not been conclusively established which form of pure magnesium stearate possesses the best lubricating properties.(36,37,41–43) Commercial lots of magnesium stearate generallyconsist of mixtures of crystalline forms.(37,39,41,42,44–46) Because of the possibility of conversion of crystalline forms during heating, consideration should be given to the pretreatment conditions employed when determining physical properties of magnesium stearate powders such as surface area.(47,48) Physical properties of magnesium stearate can vary among batches from different manufacturers(46) because the solid-state characteristics of the powder are influenced by manufacturing variables.(36) Variations in the physical properties of different lots of magnesium stearate from the same vendor have also been observed.(46) Presumably because of these variations, it has not been possible to conclusively correlate the dissolution rate retardation with observed lubricity.(49) However, various physical properties of different batches of magnesium stearate, such as specific surface area, particle size, crystalline structure, moisture content, and fatty acid composition, have been correlated with lubricant efficacy.(37,41,45,46,50–55) Due to variations in the specific surface area, the labeling states that specific surface area and the method specified for its determination should be listed on the label. Reduction in dissolution caused by the effects of magnesium stearate in some cases can be overcome by including a highly swelling disintegrant in the formulation.(56) The impact of magnesium stearate levels on tablet compaction properties and performance of roller compacted granulations has been examined.(57–59) In other compaction studies performed with granules, magnesium stearate has been shown to exert an influence on granule relaxation and may help to prevent capping.(60) There is evidence to suggest that the hydrophobic nature of magnesium stearate can vary from batch to batch owing to the presence of water-soluble, surface-active impurities such as sodium stearate. Batches containing very low concentrations of these impurities have been shown to retard the dissolution of a drug to a greater extent than when using batches that contain higher levels of impurities.(49) One study related lubricity to the fatty acid composition (stearate : palmitate) of lubricant lots for tablet formulations based on compaction data and tablet material properties.(54) However, other studies have indicated that fatty acid composition has no influence on lubricant activity(37) and highpurity magnesium stearate was as effective a lubricant as the commercial material.(10) Moisture sorption at different relative humidities can result in morphological changes in the magnesium stearate.(61,62) Magnesium stearate has been investigated for use in inhalation powders to control their performance.(63) A specification for magnesium stearate is included in the Food Chemicals Codex (FCC).(64) The EINECS number for magnesium stearate is 209-150-3.