

Name: Maleic Acid
CAS No: Z-But-2-enedioic acid [110-16-7]
BP: Maleic Acid PhEur: Maleic Acid USP-NF: Maleic Acid
Acidum maleicum; cis-butenedioic acid; cis-2-butenedioic acid; (Z)- 2-butenedioic acid; cis-ethene-1,2-dicarboxylic acid; cis-1,2-ethylenedicarboxylic acid; cis-maleic acid; maleinic acid; toxilic acid.
Z-But-2-enedioic acid [110-16-7]
C4H4O4 116.07
Maleic acid is used in the pharmaceutical industry as a pH modifier and a buffering agent.(1–3) It is also used to prevent rancidity of oils and fats; a ratio of 1 : 10 000 is usually sufficient to retard rancidity. Maleic acid is commonly used as a pharmaceutical intermediate to form the maleate salts of several categories of therapeutic agents, such as salts of antihistamines and other drug substances
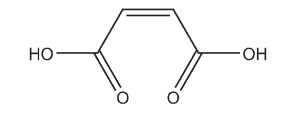
Maleic acid occurs as a white crystalline (monoclinic) powder and possesses a faint acidulous odor and an astringent taste. Fumaric acid and maleic acid are the simplest unsaturated carboxylic diacids. These acids experience two-step dissociation in aqueous solutions.(4) They have the same structural formula but different spatial configurations. Fumaric acid is the trans and maleic acid the cis isomer. The physical properties of maleic acid and fumaric acid are very different. The cis isomer is less stable. Maleic acid is used in the preparation of fumaric acid by catalytic isomerization.
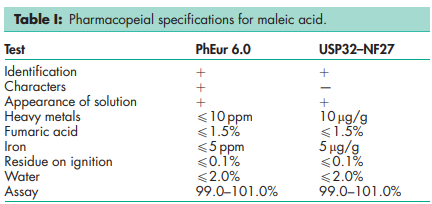
See Table I.
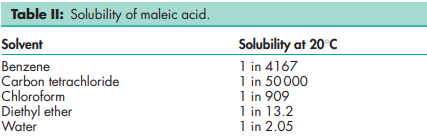
Acidity/alkalinity pH 2 (5% w/v aqueous solution at 258C) Boiling point 1358C (with decomposition) Dissociation constants pKa1 = 1.91; pKa2 = 6.33. Heat of combustion 1356.3 kJ/mol (324.2 kcal/mol) Melting point 130–1348C Partition coefficient log Kow = 0.48 (octanol/water) Solubility see Table II.(5) Specific gravity 1.590 (208C)
Maleic acid converts into the much higher-melting fumaric acid (mp: 2878C) when heated to a temperature slightly above its melting point.(6) Maleic acid is combustible when exposed to heat or flame. The bulk material should be stored in airtight glass containers and protected from light. It is recommended not to store it above 258C.
Maleic acid can react with oxidizing materials. Aqueous solutions are corrosive to carbon steels.
Maleic anhydride is the main source of maleic acid produced by hydration. Maleic anhydride is prepared commercially by the oxidation of benzene or by the reaction of butane with oxygen in the presence of a vanadium catalyst.
Maleic acid is generally regarded as a nontoxic and nonirritant material when used at low levels as an excipient. Maleic acid is used in oral, topical, and parenteral pharmaceutical formulations in addition to food products. LD50 (mouse, oral): 2.40 g/kg(7) LD50 (rabbit, skin): 1.56 g/kg LD50 (rat, oral): 0.708 g/kg
Observe normal precautions appropriate to the circumstances and quantity of the material handled. Maleic acid is considered very hazardous in the case of eye contact, which can result in corneal damage. It is also hazardous with respect to skin contact and inhalation. Skin contact can produce inflammation and blistering, with the amount of tissue damage dependent on the length of contact. Gloves, eye protection, and approved or certified respirators should be employed.
Included in the FDA Inactive Ingredients Database (IM and IV injections; oral tablets and capsules; topical applications). Included in nonparenteral and parenteral medicines licensed in the UK
Fumaric acid.
Copolymers of maleic acid (butyl monoester of poly[methylvinyl ether/maleic acid]) have found topical applications in medicated nail lacquer and mosquito repellent as film-forming agents. Compositions of certain maleic acid copolymers have been employed for preventing the attachment of dental plaque to the surface of teeth. The EINECS number for maleic acid is 203-742-5. The PubChem Compound ID (CID) for maleic acid is 444266.