

Name: Maltitol
CAS No: 4-O-a-D-Glucopyranosyl-D-glucitol [585-88-6]
BP: Maltitol PhEur: Maltitol USP-NF: Maltitol
Amalty; C*PharmMaltidex; E965; hydrogenated maltose; Malbit; Maltisorb; Maltit; D-maltitol; maltitolum.
4-O-a-D-Glucopyranosyl-D-glucitol [585-88-6]
C12H24O11 344.32
Maltitol is widely used in the pharmaceutical industry in the formulation of oral dosage forms. It is a noncariogenic bulk sweetener, approximately as sweet as sucrose, well adapted as a diluent for different oral dosage forms, wet granulation, and sugarfree hard coating.
Maltitol occurs as a white, odorless, sweet, anhydrous crystalline powder. It is a disaccharide consisting of one glucose unit linked with one sorbitol unit via an a-(1!4) bond. The crystal structure is orthorhombic.
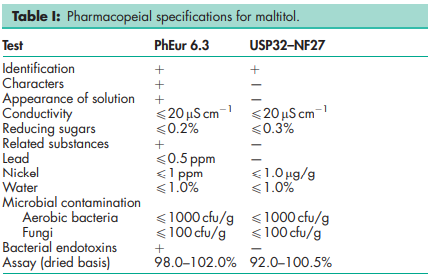
See Table I.
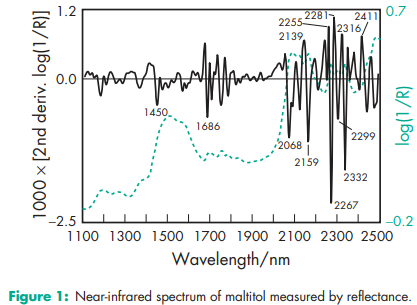
Compressibility 9.5% Density (bulk) 0.79 g/cm3 (1) Density (crystal) 1.6238 (calculated from crystallographic data).(2) Density (tapped) 0.95 g/cm3 (1) Flowability 5 seconds(1) Melting point 148–1518C NIR spectra see Figure 1. Particle size distribution 95% 4 500 mm, 40% 5 100 mm in size for Maltisorb P200 (Roquette); 95% 4 200 mm, 50% 5 100 mm in size for Maltisorb P90 (Roquette). Solubility Freely soluble in water. See also Table II. Viscosity (dynamic)

Maltitol has good thermal and chemical stability. When it is heated at temperatures above 2008C, decomposition begins (depending on time, temperature, and other prevailing conditions). Maltitol does not undergo browning reactions with amino acids, and absorbs atmospheric moisture only at relative humidities of 89% and above, at 208C.


Maltitol is obtained from hydrogenated maltose syrup. Starch is hydrolyzed to yield a high-concentration maltose syrup, which is hydrogenated with a catalyst. After purification and concentration, the syrup is crystallized.
Maltitol is used in oral pharmaceutical formulations, confectionery, and food products, and is considered to be noncariogenic. It is generally regarded as a nontoxic, nonallergenic, and nonirritant material. Digestion of maltitol follows two different metabolic pathways: absorption in the small intestine and fermentation in the large intestine (colon). These two metabolic pathways must thus be considered when evaluating the energy value. The hydrolysis of maltitol in the small intestine releases sorbitol and glucose. Glucose is actively transported and rapidly absorbed, whereas sorbitol absorption is passive. The nonabsorbed sorbitol and nonhydrolyzed maltitol are fermented by the microflora in the colon. The relative importance of the two absorption pathways depends on numerous individual factors and is related to the quantity of maltitol ingested. Excessive oral consumption (>50 g daily) may cause flatulence and diarrhea. Maltitol exhibits a low glycemic index and can therefore, under medical supervision, have a place in the diet of diabetic patients. The intake of maltitol must be taken into account for the calculation of the daily glucidic allowance. The WHO, in considering the safety of maltitol, did not set a value for the acceptable daily intake since the levels used in food to achieve a desired effect were not considered a hazard to health.(3,4)
Observe normal precautions appropriate to circumstances and quantity of material handled. Eye protection and gloves are recommended.
GRAS listed. Accepted for use as a food additive in Europe. Included in oral pharmaceutical formulations. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Sorbitol.
Maltitol is not fermented by oral bacteria and is neither acidogenic nor cariogenic. A specification for maltitol syrup is contained in the Food Chemicals Codex (FCC).(5) The EINECS number for maltitol is 209-567-0. The PubChem Compound ID (CID) for maltitol includes 3871 and 493591.