

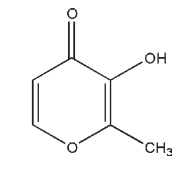
Name: Maltol
CAS No: 3-Hydroxy-2-methyl-4 H -pyran-4-one [118-71-8]
USP-NF: Maltol
3-Hydroxy-2-methyl-(1,4-pyran); 3-hydroxy-2-methyl-4-pyrone; larixinic acid; 2-methyl-3-hydroxy-4-pyrone; 2-methyl pyromeconic acid; Palatone; Veltol.
3-Hydroxy-2-methyl-4 H -pyran-4-one [118-71-8]
C6H6O3 126.11
Maltol is used in pharmaceutical formulations and food products as a flavoring agent or flavor enhancer. In foods, it is used at concentrations up to 30 ppm, particularly with fruit flavorings, although it is also used to impart a freshly baked odor and flavor to bread and cakes. When used at concentrations of 5–75 ppm, maltol potentiates the sweetness of a food product, permitting a reduction in sugar content of up to 15% while maintaining the same level of sweetness. Maltol is also used at low levels in perfumery
White crystalline solid with a characteristic, caramel-like odor and taste. In dilute solution it possesses a sweet, strawberry-like or pineapple-like flavor and odor.
See Table I.

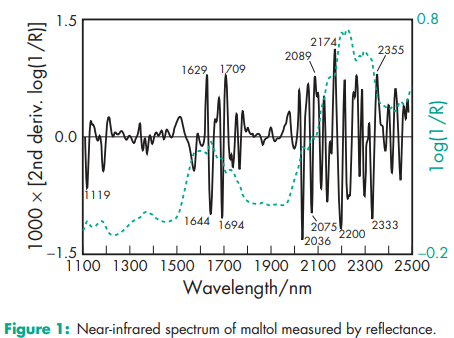
Acidity/alkalinity pH = 5.3 (0.5% w/v aqueous solution) Melting point 162–1648C (begins to sublime at 938C) NIR spectra see Figure 1. Solubility see Table II.

Maltol solutions may be stored in glass or plastic containers. The bulk material should be stored in a well-closed container, protected from light, in a cool, dry place
Concentrated solutions in metal containers, including some grades of stainless steel, may discolor on storage.
Maltol is mainly isolated from naturally occurring sources such as beechwood and other wood tars; pine needles; chicory; and the bark of young larch trees. It may also be synthesized by the alkaline hydrolysis of streptomycin salts or by a number of other synthetic methods.
Maltol is generally regarded as an essentially nontoxic and nonirritant material. In animal feeding studies, it has been shown to be well tolerated with no adverse toxic, reproductive, or embryogenic effects observed in rats and dogs fed daily intakes of up to 200 mg/kg body-weight of maltol, for 2 years.(1) The WHO has set an acceptable daily intake for maltol at up to 1 mg/kg body-weight.(2,3) A case of allergic contact dermatitis, attributed to the use of maltol in a lip ointment, has been reported.(4) LD50 (chicken, oral): 3.72 g/kg(5) LD50 (guinea pig, oral): 1.41 g/kg LD50 (mouse, oral): 0.85 g/kg LD50 (mouse, SC): 0.82 g/kg LD50 (rabbit, oral): 1.62 g/kg LD50 (rat, oral): 1.41 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Maltol should be used in a wellventilated environment. Eye protection is recommended.
GRAS listed. Included in the FDA Inactive Ingredients Database (oral solutions and syrups). Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Ethyl maltol.
Maltol is a good chelating agent and various metal complexes, e.g. aluminum maltol and ferric maltol, have been investigated as potentially useful therapeutic or experimental agents.(6–9) Maltol is a constituent of Korean red ginseng.(10) A specification for maltol is included in the Food Chemicals Codex.(11) The EINECS number for maltol is 204-271-8. The PubChem Compound ID (CID) for maltol is 8369.