

Name: Benzyl Alcohol
CAS No: Benzenemethanol [100-51-6]
BP: Benzyl Alcohol JP: Benzyl Alcohol PhEur: Benzyl Alcohol USP-NF: Benzyl Alcohol
Alcohol benzylicus; benzenemethanol; a-hydroxytoluene; phenylcarbinol; phenylmethanol; a-toluenol
Benzenemethanol [100-51-6]
C7H8O 108.14
Benzyl alcohol is an antimicrobial preservative used in cosmetics, foods, and a wide range of pharmaceutical formulations,(1–4) including oral and parenteral preparations, at concentrations up to 2.0% v/v. The typical concentration used is 1% v/v, and it has been reported to be used in protein, peptide and small molecule products, although its frequency of use has fallen from 48 products in 1996, 30 products in 2001, to 15 products in 2006.(5) In cosmetics, concentrations up to 3.0% v/v may be used as a preservative. Concentrations of 5% v/v or more are employed as a solubilizer, while a 10% v/v solution is used as a disinfectant. Benzyl alcohol 10% v/v solutions also have some local anesthetic properties, which are exploited in some parenterals, cough products, ophthalmic solutions, ointments, and dermatological aerosol sprays. Although widely used as an antimicrobial preservative, benzyl alcohol has been associated with some fatal adverse reactions when administered to neonates. It is now recommended that parenteral products preserved with benzyl alcohol, or other antimicrobial preservatives, should not be used in newborn infants if at all possible; see Section 14.
A clear, colorless, oily liquid with a faint aromatic odor and a sharp, burning taste.
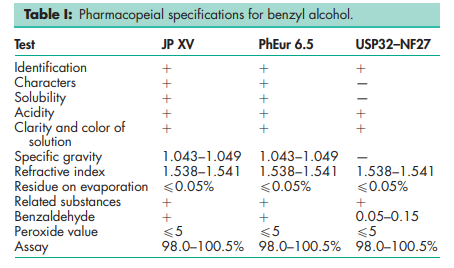
See Table I. See also Section 18.
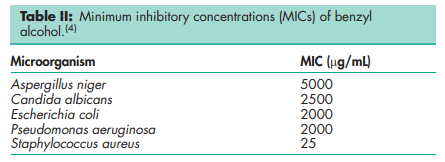
Acidity/alkalinity Aqueous solutions are neutral to litmus. Antimicrobial activity Benzyl alcohol is bacteriostatic and is used as an antimicrobial preservative against Gram-positive bacteria, molds, fungi, and yeasts, although it possesses only modest bactericidal properties. Optimum activity occurs at pH below 5; little activity is shown above pH 8. Antimicrobial activity is reduced in the presence of nonionic surfactants, such as polysorbate 80. However, the reduction in activity is less than is the case with either hydroxybenzoate esters or quaternary ammonium compounds. The activity of benzyl alcohol may also be reduced by incompatibilities with some packaging materials, particularly polyethylene; see Section 12. See Table II for reported minimum inhibitory concentrations (MICs). Bacteria Benzyl alcohol is moderately active against most Gram-positive organisms (typical MICs are 3–5 mg/mL), although some Gram-positive bacteria are very sensitive (MICs 0.025–0.05 mg/mL). In general, benzyl alcohol is less active against Gram-negative organisms. Fungi Benzyl alcohol is effective against molds and yeasts; typical MICs are 3–5 mg/mL. Spores Benzyl alcohol is inactive against spores, but activity may be enhanced by heating. Benzyl alcohol 1% v/v, at pH 5–6, has been claimed to be as effective as phenylmercuric nitrate 0.002% w/v against Bacillus stearothermophilus at 1008C for 30 min. Autoignition temperature 436.58C Boiling point 204.78C Flammability Flammable. Limits in air 1.7–15.0% v/v. Flash point 100.68C (closed cup); 104.58C (open cup). Freezing point 158C Partition coefficients Liquid paraffin : water = 0.2; Octanol : water = 1.10; Peanut oil : water = 1.3. Solubility see Table III.

Benzyl alcohol oxidizes slowly in air to benzaldehyde and benzoic acid; it does not react with water. Aqueous solutions may be sterilized by filtration or autoclaving; some solutions may generate benzaldehyde during autoclaving. Benzyl alcohol may be stored in metal or glass containers. Plastic containers should not be used; exceptions to this include polypropylene containers or vessels coated with inert fluorinated polymers such as Teflon; see Section 12. Benzyl alcohol should be stored in an airtight container, protected from light, in a cool, dry place.
Benzyl alcohol is incompatible with oxidizing agents and strong acids. It can also accelerate the autoxidation of fats. Although antimicrobial activity is reduced in the presence of nonionic surfactants, such as polysorbate 80, the reduction is less than is the case with hydroxybenzoate esters or quaternary ammonium compounds. Benzyl alcohol is incompatible with methylcellulose and is only slowly sorbed by closures composed of natural rubber, neoprene, and butyl rubber closures, the resistance of which can be enhanced by coating with fluorinated polymers.(6) However, a 2% v/v aqueous solution in a polyethylene container, stored at 208C, may lose up to 15% of its benzyl alcohol content in 13 weeks.(7) Losses to polyvinyl chloride and polypropylene containers under similar conditions are usually negligible. Benzyl alcohol can damage polystyrene syringes by extracting some soluble components.(8)
Benzyl alcohol is prepared commercially by the distillation of benzyl chloride with potassium or sodium carbonate. It may also be prepared by the Cannizzaro reaction of benzaldehyde and potassium hydroxide.
Benzyl alcohol is used in a wide variety of pharmaceutical formulations. It is metabolized to benzoic acid, which is further metabolized in the liver by conjugation with glycine to form hippuric acid, which is excreted in the urine. Ingestion or inhalation of benzyl alcohol may cause headache, vertigo, nausea, vomiting, and diarrhea. Overexposure may result in CNS depression and respiratory failure. However, the concentrations of benzyl alcohol normally employed as a preservative are not associated with such adverse effects. Reports of adverse reactions to benzyl alcohol(9,10) used as an excipient include toxicity following intravenous administration;(11–13) neurotoxicity in patients administered benzyl alcohol in intrathecal preparations;(14,15) hypersensitivity,(16–18) although relatively rare; and a fatal toxic syndrome in premature infants.(19–22) The fatal toxic syndrome in low-birth-weight neonates, which includes symptoms of metabolic acidosis and respiratory depression, was attributed to the use of benzyl alcohol as a preservative in solutions used to flush umbilical catheters. As a result of this, the FDA has recommended that benzyl alcohol should not be used in such flushing solutions and has advised against the use of medicines containing preservatives in the newborn.(23,24) The WHO has set the estimated acceptable daily intake of the benzyl/benzoic moiety at up to 5 mg/kg body-weight daily.(25) LD50 (mouse, IV): 0.32 g/kg(26) LD50 (mouse, oral): 1.36 g/kg LD50 (rat, IP): 0.4 g/kg LD50 (rat, IV): 0.05 g/kg LD50 (rat, oral): 1.23 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Benzyl alcohol (liquid and vapor) is irritant to the skin, eyes, and mucous membranes. Eye protection, gloves, and protective clothing are recommended. Benzyl alcohol should be handled in a well-ventilated environment; a selfcontained breathing apparatus is recommended in areas of poor ventilation. Benzyl alcohol is flammable.
Included in the FDA Inactive Ingredients Database (dental injections, oral capsules, solutions and tablets, topical, and vaginal preparations). Included in parenteral and nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Benzyl alcohol is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. The EINECS number for benzyl alcohol is 202-859-9. The PubChem Compound ID (CID) for benzyl alcohol is 244.