

Name: Zinc Stearate
CAS No: Octadecanoic acid zinc salt [557-05-1]
BP: Zinc Stearate PhEur: Zinc Stearate USP: Zinc Stearate
Cecavon; Demarone; dibasic zinc stearate; HyQual; Kemilub; Metallac; stearic acid zinc salt; Synpro; zinc distearate; zinc octadecanoate; zinc soap; zinci stearas
Octadecanoic acid zinc salt [557-05-1]
C36H70O4Zn 632.33 (for pure material) The USP 32 describes zinc stearate as a compound of zinc with a mixture of solid organic acids obtained from fats, and consists chiefly of variable proportions of zinc stearate and zinc palmitate. It contains the equivalent of 12.5–14.0% of zinc oxide (ZnO). The PhEur 6.0 states that zinc stearate [(C17H35COO)2Zn] may contain varying proportions of zinc palmitate [(C15H31COO)2Zn] and zinc oleate [(C17H33COO)2Zn]. It contains not less than 10.0% and not more than 12.0% of zinc.
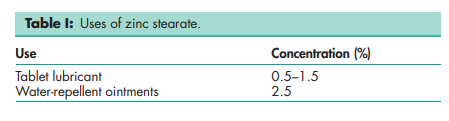
Zinc stearate is primarily used in pharmaceutical formulations as a lubricant in tablet and capsule manufacture at concentrations up to 1.5% w/w. It has also been used as a thickening and opacifying agent in cosmetic and pharmaceutical creams, and as a dusting powder. See Table I.
Zinc stearate occurs as a fine, white, bulky, hydrophobic powder, free from grittiness and with a faint characteristic odor.
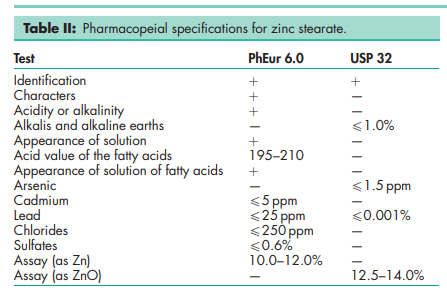
See Table II.
Autoignition temperature 4218C Density 1.09 g/cm3 Flash point 2778C Melting point 120–1228C; 1308C also reported.(1,2) Particle size distribution 100% through a 44.5-mm sieve (#325 mesh). Solubility Practically insoluble in ethanol (95%), ether, water, and oxygenated solvents; soluble in acids, benzene, and other aromatic solvents


Zinc stearate is stable and should be stored in a well-closed container in a cool, dry place.
Zinc stearate is decomposed by dilute acids. It is incompatible with strong oxidizing agents.
An aqueous solution of zinc sulfate is added to sodium stearate solution to precipitate zinc stearate. The zinc stearate is then washed with water and dried. Zinc stearate may also be prepared from stearic acid and zinc chloride.
Zinc stearate is used in oral and topical pharmaceutical formulations, and is generally regarded as a nontoxic and nonirritant excipient. However, following inhalation, it has been associated with fatal pneumonitis, particularly in infants.(1) As a result, zinc stearate has now been removed from baby dusting powders. LD50 (rat, IP): 0.25 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended. Zinc stearate may be harmful on inhalation and should be used in a well-ventilated environment; a respirator is recommended. In the UK, the long-term (8-hour TWA) workplace exposure limit for zinc stearate is 10 mg/m3 for total inhalable dust and 4 mg/m3 for respirable dust. The short-term (15-minutes) workplace exposure limit for total inhalable dust is 20 mg/m3 . (2) In the USA, the OSHA limit is 15 mg/m3 for total dust, 5 mg/m3 respirable fraction for zinc stearate.(3) When heated to decomposition, zinc stearate emits acrid smoke and fumes of zinc oxide.
GRAS listed. Included in the FDA Inactive Ingredients Database (oral capsules and tablets). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Calcium stearate; magnesium stearate; stearic acid.
The EINECS number for zinc stearate is 209-151-9.