BP: Purified Water JP: Purified Water PhEur: Water, Purified USP: Purified Water
Aqua; aqua purificata; hydrogen oxide.
Water [7732-18-5]
H2O ,18.02
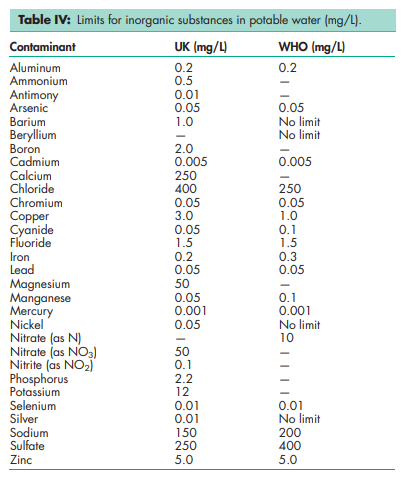
Water is widely used as a raw material, ingredient and solvent in the processing, formulation and manufacture of pharmaceutical products, active pharmaceutical ingredients (API) and intermediates, and analytical reagents. Specific grades of water are used for particular applications in concentrations up to 100%; see Table I.
The term ‘water’ is used to describe potable water that is freshly drawn direct from the public supply and is suitable for drinking. Water used in the pharmaceutical industry and related disciplines is classified as either drinking (potable) water, purified water, sterile purified water, water for injection (WFI), sterile water for injection, bacteriostatic water for injection, sterile water for irrigation, or sterile water for inhalation. Validation is required for all systems producing the water indicated, with the exception of potable water. The chemical composition of potable water is variable, and the nature and concentrations of the impurities in it depend upon the source from which it is drawn. Water classified as potable water for applications such as some initial rinsing and API manufacturing operations, must meet the US Environmental Protection Agency’s National Primary Drinking Water Regulations, or comparable regulations of the EU or Japan. For most pharmaceutical applications, potable water is purified by distillation, ion exchange treatment, reverse osmosis (RO), or some other suitable process to produce ‘purified water’. For certain applications, water with pharmacopeial specifications differing from those of purified water should be used, e.g. WFI;Water is a clear, colorless, odorless, and tasteless liquid.
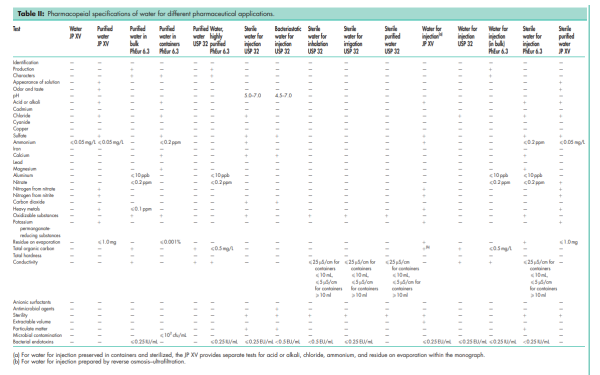
See Table II.
oiling point 1008C Critical pressure 22.1 MPa (218.3 atm) Critical temperature 374.28C Dielectric constant D 25 = 78.54 Dipole moment 1.76 in benzene at 258C; 1.86 in dioxane at 258C. Ionization constant 1.008 1014 at 258C. Latent heat of fusion 6 kJ/mol (1.436 kcal/mol) Latent heat of vaporization 40.7 kJ/mol (9.717 kcal/mol) Melting point 08C Refractive index nD 20 = 1.3330 Solubility Miscible with most polar solvents. Specific gravity 0.9971 at 258C. Specific heat (liquid) 4.184 J/g/8C (1.00 cal/g/8C) at 148C. Surface tension 71.97 mN/m (71.97 dynes/cm) at 258C. Vapor pressure 3.17 kPa (23.76 mmHg) at 258C. Viscosity (dynamic) 0.89 mPa s (0.89 cP) at 258C.
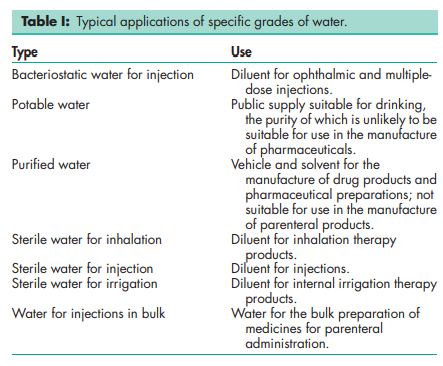
Water is chemically stable in all physical states (ice, liquid, and vapor). Water leaving the pharmaceutical purification system and entering the storage tank must meet specific requirements. The goal when designing and operating the storage and distribution system is to keep the water from exceeding allowable limits during storage. In particular, the storage and distribution system must ensure that water is protected against ionic and organic contamination, which would lead to an increase in conductivity and total organic carbon, respectively. The system must also be protected against physical entry of foreign particles and microorganisms so that microbial growth is prevented or minimized. Water for specific purposes should be stored in appropriate containers; see Table III.
In pharmaceutical formulations, water can react with drugs and other excipients that are susceptible to hydrolysis (decomposition in the presence of water or moisture) at ambient and elevated temperatures. Water can react violently with alkali metals and rapidly with alkaline metals and their oxides, such as calcium oxide and magnesium oxide. Water also reacts with anhydrous salts to form hydrates of various compositions, and with certain organic materials and calcium carbide.
Unlike other excipients, water is not purchased from outside suppliers but is manufactured in-house by pharmaceutical companies. As naturally occurring water has a variety of contaminants, many treatment processes have been developed to remove these. A typical pharmaceutical water purification system contains several unit operations designed to remove various components. The selection of the most appropriate system and its overall design are crucial factors in ensuring that water of the correct quality is produced.(1,2) To produce potable or drinking water, insoluble matter is first removed from a water supply by coagulation, settling (clarification), and filtering processes. Pathogenic microorganisms present are then destroyed by aeration, chlorination, or some other means. Water may also be rendered free of viable pathogenic microorganisms by active boiling for 15–20 minutes. Activated carbon filters are employed to remove chlorine and many dissolved organic materials found in water, although they may become a breeding ground for microorganisms. The palatability of the water is improved by aeration and charcoal filtration. Purified water suitable for use in pharmaceutical formulations is usually prepared by purifying potable water by one of several processes, such as distillation, deionization, or RO.(1,3–8) The quality attributes of WFI are stricter than those for purified water. Consequently, the preparation methods typically vary in the last stage to ensure good control of WFI quality. Methods for the production of WFI are the subject of current debate. The PhEur 6.3 indicates that only distillation would give assurance of consistent supply of the appropriate quality, but permits distillation, ion exchange, RO or any other suitable method that complies with regulations on water intended for human consumption laid down by the competent authority. The USP 32 and the JP XV permit the use of RO in addition to distillation and ultrafiltration. In the past 10–15 years, RO has become the most common way to produce pharmaceutical purified water, either as a final treatment step or as a pretreatment step for the distillation stills. Distillation Distillation is a process that involves the evaporation of water followed by the condensation of the resulting steam. While expensive, it allows removal of almost all organic and inorganic impurities and achieves very high quality water. It is also considered the safest method to avoid microbial and endotoxin contamination. To improve energy efficiency, distillation is usually conducted in multiple-effects stills designed to recover most of the energy spent on evaporating the water. A typical design consists of an evaporator, vapor separator, and compressor. The distilland (raw feed water) is heated in the evaporator to boiling and the vapor produced is separated from entrained distilland in the separator. The vapor then enters a compressor where the temperature of the vapors is raised to 1078C. Superheated vapors are then condensed on the outer surface of the tubes of the evaporator containing cool distilland circulating within. Vapor compression stills of various sizes are commercially available and can be used to produce water of high purity when properly constructed. A high-quality distillate, such as WFI, can be obtained if the water is first deionized. The best stills are constructed from types 304 or 316 stainless steel and coated with pure tin, or are made from chemical-resistant glass. Deionization An ionic exchange process is based on the ability of certain synthetic resins to selectively adsorb either cations or anions, and to release (exchange) other ions based on their relative activity. Cationic and anionic ion exchange resins are used to purify potable water by removing any dissolved ions. Dissolved gases are also removed, while chlorine, in the concentrations generally found in potable water, is destroyed by the resin itself. Some organics and colloidal particles are removed by adsorption and filtration. Resin beds may, however, foster microbial life and produce pyrogenic effluent unless adequate precautions are taken to prevent contamination. Another disadvantage is the type of chemicals required for resin regeneration. A continuous deionization system, which represents a combination of ion exchange and membrane separation technologies, uses an electrical current to continuously regenerate the ion exchange resin simultaneously with the water treatment process, eliminating the need to handle powerful chemicals. Ion exchange units are normally used today to treat raw feed water prior to distillation or RO processing. Reverse osmosis Water is forced through a semipermeable membrane in the opposite direction to normal osmotic diffusion. Typically, membranes range between 1–10 A˚ and reject not only organic compounds, bacteria and viruses, but also 90–99% of all ions. It is common to use double-pass RO systems with two filtration stages connected in series. Such systems meet requirements for USP purified water and WFI. However, EU regulations do not allow RO to be used as a final treatment step for the production of WFI. Membrane filtration Membrane filters are surface-type filters, which stop particles larger than the pore size at the upstream surface of the polymeric membrane. Microfiltration uses membranes with pores in the 0.1–1.0 mm range, which can filter out particles of dust, activated carbon, ion exchange resin fines, and most microorganisms. Ultrafiltration uses membranes that reject not only solid particles but also dissolved matter with a high molecular weight. The ‘molecular weight cut-off’ point of such membranes vary in the range 10 000–100 000 Da, and bacteria, endotoxins, colloidal contaminants, and large organic molecules can be removed.
Water is the base for many biological life forms, and its safety in pharmaceutical formulations is unquestioned provided it meets standards of quality for potability(9) and microbial content; see Sections 9 and 18. Plain water is considered slightly more toxic upon injection into laboratory animals than physiological salt solutions such as normal saline or Ringer’s solution. Ingestion of excessive quantities of water can lead to water intoxication, with disturbances of the electrolyte balance. WFI should be free from pyrogens. LD50 (mouse, IP): 25 g/kg(10)
Observe normal precautions appropriate to the circumstances and quantity of material handled.
Included in non-parenteral and parenteral medicines licensed in the UK and USA.
Bacteriostatic water for injection; carbon dioxide-free water; deaerated water; hard water; soft water; sterile water for inhalation; sterile water for injection; sterile water for irrigation; water for injection (WFI). Bacteriostatic water for injection Comments The USP 32 describes bacteriostatic water for injection as sterile water for injection that contains one or more suitable antimicrobial agents. Carbon dioxide-free water Comments Purified water that has been boiled vigorously for 5 minutes and allowed to cool while protecting it from absorption of atmospheric carbon dioxide. De-aerated water Comments Purified water that has been boiled vigorously for 5 minutes and cooled to reduce the air (oxygen) content. Hard water Comments Water containing the equivalent of not less than 120 mg/L and not more than 180 mg/L of calcium carbonate. Soft water Comments Water containing the equivalent of not more than 60 mg/L of calcium carbonate. Sterile water for inhalation Comments The USP 32 describes sterile water for inhalation as WFI sterilized and suitably packaged. It contains no antimicrobial agents or other added substances, except where used in humidifiers or other similar devices, and where liable to contamination over a period of time. Sterile water for injection Comments The USP 32 describes sterile water for injection as WFI sterilized and suitably packaged. It contains no antimicrobial agents or other substances. Sterile water for injection in containers is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. Sterile water for irrigation Comments The USP 32 describes sterile water for irrigation as WFI sterilized and suitably packaged. It contains no antimicrobial agents or other substances. Water for injection (WFI) Comments The USP 32 describes WFI as water purified by distillation or RO. It contains no added substances. The PhEur 6.3 title is ‘water for injections’ and comprises two parts: ‘water for injections in bulk’ and ‘sterilized water for injection’. The PhEur 6.3 states that water for injections is produced by distillation.
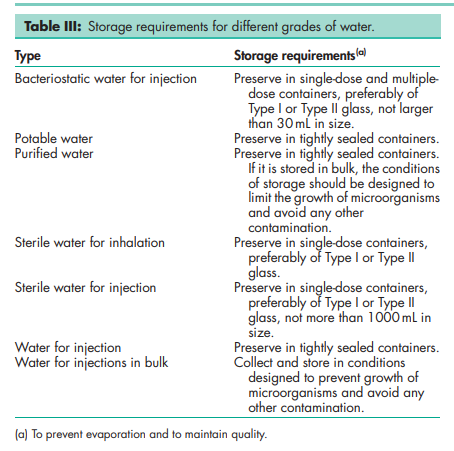
In most pharmacopeias, the term ‘water’ now refers to purified or distilled water. Without further purification, ‘water’ may be unsuitable for certain pharmaceutical applications; for example, the presence of calcium in water affects the viscosity and gel strength of algins and pectin dispersions, while the use of potable water affects the clarity and quality of cough mixtures, and the stability of antibiotic liquid preparations. Water commonly contains salts of aluminum, calcium, iron, magnesium, potassium, sodium, and zinc. Toxic substances such as arsenic, barium, cadmium, chromium, cyanide, lead, mercury, and selenium may constitute a danger to health if present in excessive amounts. Ingestion of water containing high amounts of calcium and nitrate is also contraindicated. National standards generally specify the maximum limits for these inorganic substances in potable water. Limits have also been placed on microorganisms, detergents, phenolics, chlorinated phenolics, and other organic substances. The WHO(11) and national bodies have issued guidelines for water quality, although many countries have their own standards for water quality embodied in specific legislation.(12) Control of microbiological contamination is critical for waters used in preparation of pharmaceuticals, as proliferation of microorganisms can potentially occur during all stages of manufacture, storage, or distribution. Suitable control is achieved by ensuring that the water system is well designed and well maintained. Purified water that is produced, stored, and circulated at ambient temperatures is susceptible to the establishment of biofilms; therefore, frequent monitoring, high usage, correct flow rate, and appropriate sanitization are all factors that require consideration to ensure that water is satisfactory.(13) Monitoring of the whole system is essential in order to demonstrate that correct microbiological quality is achieved. For WFI, the recommended methodology is membrane filtration (0.45 mm) as a large sample size (100–300 mL) is required. For purified water, membrane filtration or plate count methods are typically used depending on the quality requirements of the system. It is important to set appropriate target, alert, and action limits to serve as an indication of action required to bring the quality of water back under control. It is recognized that limits are not intended as pass/fail criteria for water or product batches; however, an investigation regarding the implications should be conducted.(14) Validation is conducted to provide a high level of assurance that the water production and distribution system will consistently produce water conforming to a defined quality specification. The validation process serves to qualify the design (DQ), installation (IQ), operation (OQ), and performance (PQ) of the system. The extent of monitoring data required should be defined, with consideration given to whether validation to FDA guidelines is required.(14) It is also important to have an ongoing control program with respect to maintenance, and periodic reviews of the performance of the water system. The PubChem Compound ID (CID) for water is 962. See Table IV.