

Name: Mannitol
CAS No: D-Mannitol [69-65-8]
BP: Mannitol JP: D-Mannitol PhEur: Mannitol USP: Mannitol
Cordycepic acid; C*PharmMannidex; E421; Emprove; manna sugar; D-mannite; mannite; mannitolum; Mannogem; Pearlitol.
D-Mannitol [69-65-8]
C6H14O6 182.17
Mannitol is widely used in pharmaceutical formulations and food products. In pharmaceutical preparations it is primarily used as a diluent (10–90% w/w) in tablet formulations, where it is of particular value since it is not hygroscopic and may thus be used with moisture-sensitive active ingredients.(1,2) Mannitol may be used in direct-compression tablet applications,(3,4) for which the granular and spray-dried forms are available, or in wet granulations.(5,6) Granulations containing mannitol have the advantage of being dried easily. Specific tablet applications include antacid preparations, glyceryl trinitrate tablets, and vitamin preparations. Mannitol is commonly used as an excipient in the manufacture of chewable tablet formulations because of its negative heat of solution, sweetness, and ‘mouth feel’. (6,7) In lyophilized preparations, mannitol (20–90% w/w) has been included as a carrier to produce a stiff, homogeneous cake that improves the appearance of the lyophilized plug in a vial.(8–10) A pyrogen-free form is available specifically for this use. Mannitol has also been used to prevent thickening in aqueous antacid suspensions of aluminum hydroxide (<7% w/v). It has been suggested as a plasticizer in soft-gelatin capsules, as a component of sustained-release tablet formulations,(11) and as a carrier in dry powder inhalers.(12,13) It is also used as a diluent in rapidly dispersing oral dosage forms.(14,15) It is used in food applications as a bulking agent. Therapeutically, mannitol administered parenterally is used as an osmotic diuretic, as a diagnostic agent for kidney function, as an adjunct in the treatment of acute renal failure, and as an agent to reduce intracranial pressure, treat cerebral edema, and reduce intraocular pressure. Given orally, mannitol is not absorbed significantly from the gastrointestinal tract, but in large doses it can cause osmotic diarrhea
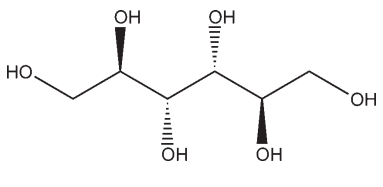
Mannitol is D-mannitol. It is a hexahydric alcohol related to mannose and is isomeric with sorbitol. Mannitol occurs as a white, odorless, crystalline powder, or freeflowing granules. It has a sweet taste, approximately as sweet as glucose and half as sweet as sucrose, and imparts a cooling sensation in the mouth. Microscopically, it appears as orthorhombic needles when crystallized from alcohol. Mannitol shows polymorphism.
See Table I
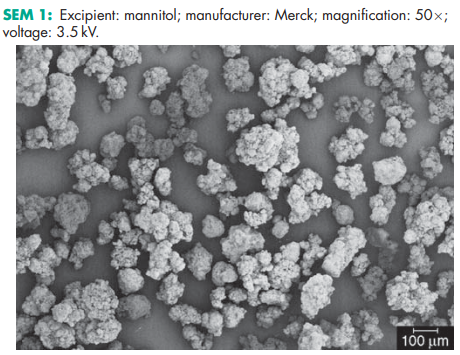
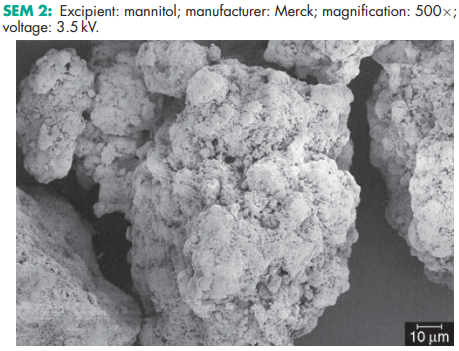


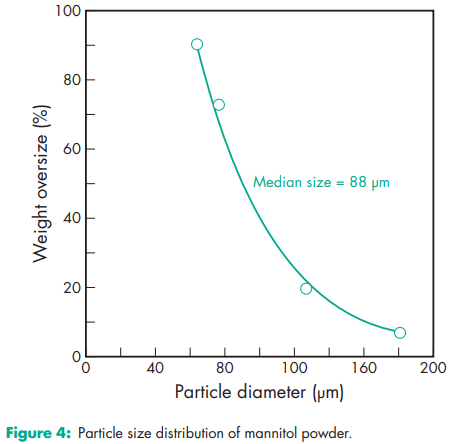
Compressibility see Figure 1. Density (bulk) 0.430 g/cm3 for powder; 0.7 g/cm3 for granules. Density (tapped) 0.734 g/cm3 for powder; 0.8 g/cm3 for granules. Density (true) 1.514 g/cm3 Dissociation constant pKa = 13.5 at 188C Flash point <1508C Flowability Powder is cohesive, granules are free flowing. Heat of combustion 16.57 kJ/g (3.96 kcal/g) Heat of solution 120.9 J/g (28.9 cal/g) at 258C Melting point 166–1688C Moisture content see Figure 2. NIR spectra see Figure 3. Osmolarity A 5.07% w/v aqueous solution is isoosmotic with serum. Particle size distribution Pearlitol 300 DC: maximum of 0.1% greater than 500 mm and minimum of 90% greater than 200 mm in size; Pearlitol 400 DC: maximum of 20% greater than 500 mm and minimum of 85% greater than 100 mm in size; Pearlitol 500 DC: maximum of 0.5% greater than 841 mm and minimum of 90% greater than 150 mm in size. Average particle diameter is 250 mm for Pearlitol 300 DC, 360 mm for Pearlitol 400 DC and 520 mm for Pearlitol 500 DC. (17) See also Figure 4. Refractive index nD 20 = 1.333 Solubility see Table II. Specific surface area 0.37–0.39 m2 /g
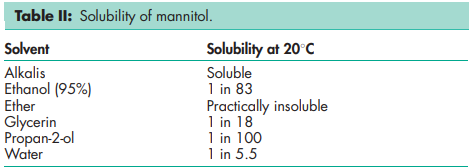
Mannitol is stable in the dry state and in aqueous solutions. Solutions may be sterilized by filtration or by autoclaving and if necessary may be autoclaved repeatedly with no adverse physical or chemical effects.(18) In solution, mannitol is not attacked by cold, dilute acids or alkalis, nor by atmospheric oxygen in the absence of catalysts. Mannitol does not undergo Maillard reactions. The bulk material should be stored in a well-closed container in a cool, dry placeoquette Fre`res). Tablet diameter: 20 mm. Lubricant: magnesium stearate 0.7% w/w for Pearlitol 400 DC and Pearlitol 500 DC; magnesium stearate 1% w/w for Pearlitol 300 DC.
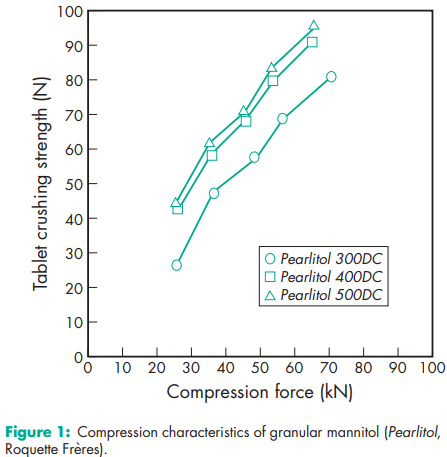
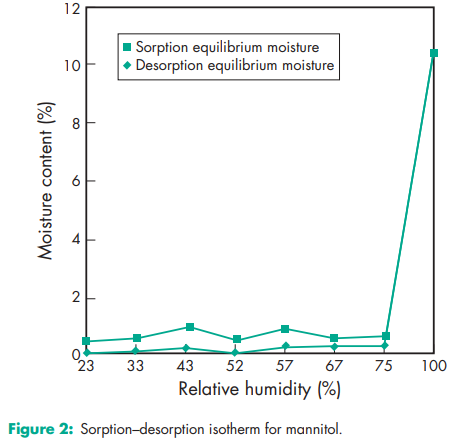
Mannitol solutions, 20% w/v or stronger, may be salted out by potassium chloride or sodium chloride.(19) Precipitation has been reported to occur when a 25% w/v mannitol solution was allowed to contact plastic.(20) Sodium cephapirin at 2 mg/mL and 30 mg/mL concentration is incompatible with 20% w/v aqueous mannitol solution. Mannitol is incompatible with xylitol infusion and may form complexes with some metals such as aluminum, copper, and iron. Reducing sugar impurities in mannitol have been implicated in the oxidative degradation of a peptide in a lyophilized formation.(21) Mannitol was found to reduce the oral bioavailability of cimetidine compared to sucrose.(22)
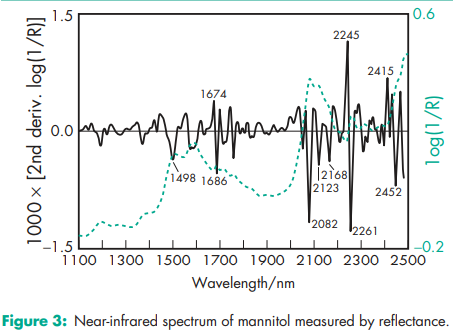
Mannitol may be extracted from the dried sap of manna and other natural sources by means of hot alcohol or other selective solvents. It is commercially produced by the catalytic or electrolytic reduction of monosaccharides such as mannose and glucose.
Mannitol is a naturally occurring sugar alcohol found in animals and plants; it is present in small quantities in almost all vegetables. Laxative effects may occur if mannitol is consumed orally in large quantities.(23) If it is used in foods as a bodying agent and daily ingestion of over 20 g is foreseeable, the product label should bear the statement ‘excessive consumption may have a laxative effect’. After intravenous injection, mannitol is not metabolized to any appreciable extent and is minimally reabsorbed by the renal tubule, about 80% of a dose being excreted in the urine in 3 hours.(24) A number of adverse reactions to mannitol have been reported, primarily following the therapeutic use of 20% w/v aqueous intravenous infusions.(25) The quantity of mannitol used as an excipient is considerably less than that used therapeutically and is consequently associated with a lower incidence of adverse reactions. However, allergic, hypersensitive-type reactions may occur when mannitol is used as an excipient. An acceptable daily intake of mannitol has not been specified by the WHO since the amount consumed as a sweetening agent was not considered to represent a hazard to health.(26) LD50 (mouse, IP): 14 g/kg(27) LD50 (mouse, IV): 7.47 g/kg LD50 (mouse, oral): 22 g/kg LD50 (rat, IV): 9.69 g/kg LD50 (rat, oral): 13.5 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Mannitol may be irritant to the eyes; eye protection is recommended.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (IP, IM, IV, and SC injections; infusions; buccal, oral and sublingual tablets, powders and capsules; ophthalmic preparations; topical solutions). Included in nonparenteral and parenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Mon-medicinal Ingredients.
Sorbitol.
Mannitol is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. Mannitol is an isomer of sorbitol, the difference between the two polyols occurring in the planar orientation of the OH group on the second carbon atom. Each isomer is characterized by its own individual set of properties, the most important difference being the response to moisture. Sorbitol is hygroscopic, while mannitol resists moisture sorption, even at high relative humidities. Granular mannitol flows well and imparts improved flow properties to other materials. However, it usually cannot be used with concentrations of other materials exceeding 25% by weight. Recommended levels of lubricant are 1% w/w calcium stearate or 1–2% w/w magnesium stearate. Suitable binders for preparing granulations of powdered mannitol are gelatin, methylcellulose 400, starch paste, povidone, and sorbitol. Usually, 3–6 times as much magnesium stearate or 1.5–3 times as much calcium stearate is needed for lubrication of mannitol granulations than is needed for other excipients. A study has examined the influence of common excipients such as sucrose and trehalose, on the crystallization of mannitol in freezedrying.(28) Mannitol has been reported to sublime at 1308C.(29) Ludiflash (BASF) is a coprocessed excipient used as a tablet filler, binder, and disintegrant, and contains mainly mannitol, and also crospovidone and polyvinyl acetate. A specification for mannitol is contained in the Food Chemicals Codex (FCC).(30) The EINECS number for mannitol is 200-711-8. The PubChem Compound ID (CID) for mannitol includes 6251 and 453.