

Name: Hydroxyethyl Cellulose
CAS No: Cellulose, 2-hydroxyethyl ether [9004-62-0]
BP: Hydroxyethylcellulose PhEur: Hydroxyethylcellulose USP-NF: Hydroxyethyl Cellulose
Cellosize HEC; cellulose hydroxyethyl ether; cellulose 2-hydroxyethyl ether; cellulose hydroxyethylate; ethylhydroxy cellulose; ethylose; HEC; HE cellulose; hetastarch; 2-hydroxyethyl cellulose ether; hydroxyethylcellulosum; hydroxyethyl ether cellulose; hydroxyethyl starch; hyetellose; Natrosol; oxycellulose; Tylose H; Tylose PHA.
Cellulose, 2-hydroxyethyl ether [9004-62-0]
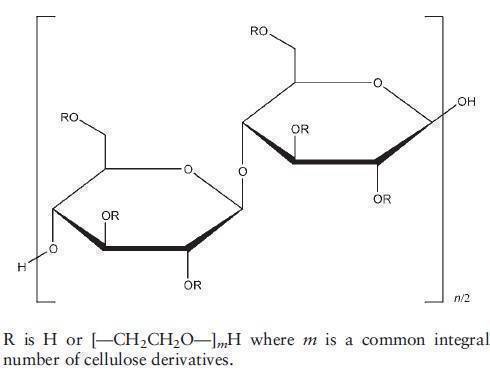
The USP32-NF27 describes hydroxyethyl cellulose as a partially substituted poly(hydroxyethyl) ether of cellulose. It is available in several grades that vary in viscosity and degree of substitution; some grades are modified to improve their dispersion in water. The grades are distinguished by appending a number indicative of the apparent viscosity, in mPa s, of a 2% w/v solution measured at 208C. Hydroxyethyl cellulose may also contain a suitable anticaking agent.
Hydroxyethyl cellulose is a nonionic, water-soluble polymer widely used in pharmaceutical formulations. It is primarily used as a thickening agent in ophthalmic(1) and topical formulations,(2) although it is also used as a binder(3) and film-coating agent for tablets.(4) It is present in lubricant preparations for dry eye, contact lens care, and dry mouth.(5) The concentration of hydroxyethyl cellulose used in a formulation is dependent upon the solvent and the molecular weight of the grade. Hydroxyethyl cellulose is also widely used in cosmetics.
Hydroxyethyl cellulose occurs as a white, yellowish-white or grayish-white, odorless and tasteless, hygroscopic powder.
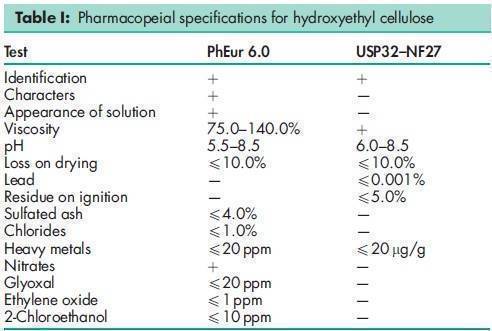
See Table I.
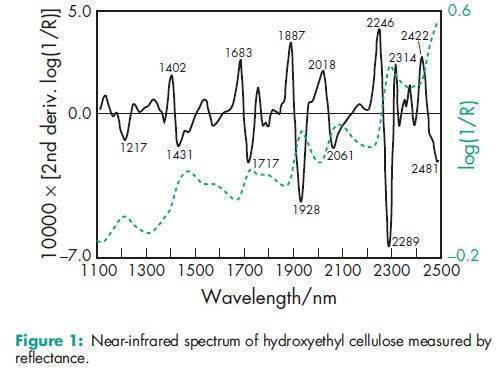
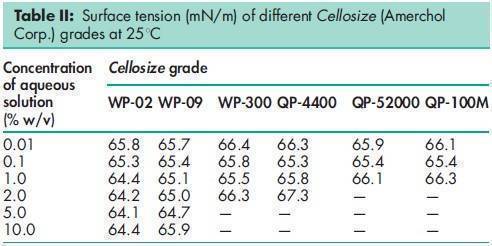
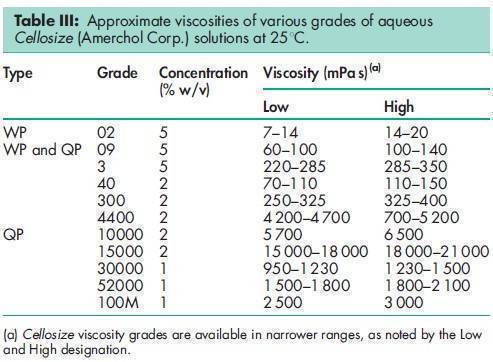
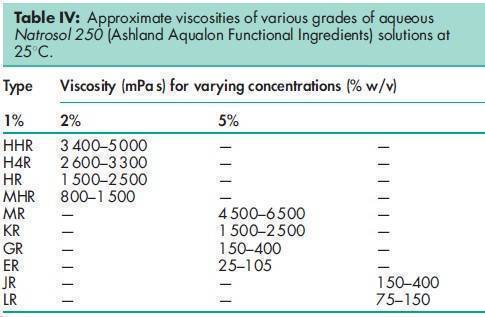
Acidity/alkalinity pH = 5.5–8.5 for a 1% w/v aqueous solution. Ash 2.5% w/w for Cellosize; 3.5% w/w for Natrosol. Autoignition temperature 4208C Density (bulk) 0.35–0.61 g/cm3 for Cellosize; 0.60 g/cm3 for Natrosol. Melting point Softens at 135–1408C; decomposes at about 2808C. Moisture content Commercially available grades of hydroxyethyl cellulose contain less than 5% w/w of water. However, as hydroxyethyl cellulose is hygroscopic, the amount of water absorbed depends upon the initial moisture content and the relative humidity of the surrounding air. Typical equilibrium moisture values for Natrosol 250 at 258C are: 6% w/w at 50% relative humidity and 29% w/w at 84% relative humidity. NIR spectra see Figure 1. Particle size distribution Cellosize: 100% through a US #80 mesh (177 mm); Natrosol (regular grind): 10% retained on a US #40 mesh (420 mm); Natrosol (X-grind): 0.5% retained on a US #60 mesh (250 mm). Refractive index n D 20 = 1.336 for a 2% w/v aqueous solution. Solubility Hydroxyethyl cellulose is soluble in either hot or cold water, forming clear, smooth, uniform solutions. Practically insoluble in acetone, ethanol (95%), ether, toluene, and most other organic solvents. It is nonionic. In some polar organic solvents, such as the glycols, hydroxyethyl cellulose either swells or is partially soluble. Specific gravity 1.38–1.40 for Cellosize; 1.0033 for a 2% w/v aqueous hydroxyethyl cellulose solution. Surface tension see Table II. Viscosity (dynamic) Hydroxyethyl cellulose is available in a wide range of viscosity types; e.g. Cellosize is manufactured in 11 regular viscosity grades. Hydroxyethyl cellulose grades differ principally in their aqueous solution viscosities which range from 2–20 000 mPa s for a 2% w/v aqueous solution. Two types of Cellosize are produced, a WP-type, which is a normaldissolving material, and a QP-type, which is a rapid-dispersing material. The lowest viscosity grade (02) is available only in the WP-type. Five viscosity grades (09, 3, 40, 300, and 4400) are produced in both WP- and QP-types. Five high-viscosity grades (10000, 15000, 30000, 52000, and 100M) are produced only in the QP-type. For the standard Cellosize grades and types available and their respective viscosity ranges in aqueous solution, see Table III. Natrosol 250 has a degree of substitution of 2.5 and is produced in 10 viscosity types. The suffix ‘R’ denotes that Natrosol has been surface-treated with glyoxal to aid in solution preparation; see Table IV. Aqueous solutions made using a rapidly dispersing material may be prepared by dispersing the hydroxyethyl cellulose in mildly agitated water at 20–258C. When the hydroxyethyl cellulose has been thoroughly wetted, the temperature of the solution may be increased to 60–708C to increase the rate of dispersion. Making the solution slightly alkaline also increases the dispersion process. Typically, complete dispersion may be achieved in approximately an hour by controlling the temperature, pH, and rate of stirring. Normally dispersing grades of hydroxyethyl cellulose require more careful handling to avoid agglomeration during dispersion; the water should be stirred vigorously. Alternatively, a slurry of hydroxyethyl cellulose may be prepared in a nonaqueous solvent, such as ethanol, prior to dispersion in water.
Hydroxyethyl cellulose powder is a stable though hygroscopic material. Aqueous solutions of hydroxyethyl cellulose are relatively stable at pH 2–12 with the viscosity of solutions being largely unaffected. However, solutions are less stable below pH 5 owing to hydrolysis. At high pH, oxidation may occur. Increasing the temperature reduces the viscosity of aqueous hydroxyethyl cellulose solutions. However, on cooling, the original viscosity is restored. Solutions may be subjected to freeze–thawing, high-temperature storage, or boiling without precipitation or gelation occurring. Hydroxyethyl cellulose is subject to enzymatic degradation, with consequent loss in viscosity of its solutions.(6) Enzymes that catalyze this degradation are produced by many bacteria and fungi present in the environment. For prolonged storage, an antimicrobial preservative should therefore be added to aqueous solutions. Aqueous solutions of hydroxyethyl cellulose may also be sterilized by autoclaving. Hydroxyethyl cellulose powder should be stored in a well-closed container, in a cool, dry place.
Hydroxyethyl cellulose is insoluble in most organic solvents. It is incompatible with zein and partially compatible with the following water-soluble compounds: casein; gelatin; methylcellulose; poly-vinyl alcohol, and starch. Hydroxyethyl cellulose can be used with a wide variety of water-soluble antimicrobial preservatives. However, sodium pentachlorophenate produces an immediate increase in viscosity when added to hydroxyethyl cellulose solutions. Hydroxyethyl cellulose has good tolerance for dissolved electrolytes, although it may be salted out of solution when mixed with certain salt solutions. For example, the following salt solutions will precipitate a 10% w/v solution of Cellosize WP-09 and a 2% w/v solution of Cellosize WP-4400: sodium carbonate 50% and saturated solutions of aluminum sulfate; ammonium sulfate; chromic sulfate; disodium phosphate; magnesium sulfate; potassium ferrocyanide; sodium sulfate; sodium sulfite; sodium thiosulfate; and zinc sulfate. Natrosol is soluble in most 10% salt solutions, excluding sodium carbonate and sodium sulfate, and many 50% salt solutions with the exception of the following: aluminum sulfate; ammonium sulfate; diammonium phosphate; disodium phosphate; ferric chloride; magnesium sulfate; potassium ferrocyanide; sodium metaborate; sodium nitrate; sodium sulfite; trisodium phosphate; and zinc sulfate. Natrosol 150 is generally more tolerant of dissolved salts than is Natrosol 250. Hydroxyethyl cellulose is also incompatible with certain fluorescent dyes or optical brighteners, and certain quaternary disinfectants which will increase the viscosity of aqueous solutions.
A purified form of cellulose is reacted with sodium hydroxide to produce a swollen alkali cellulose, which is chemically more reactive than untreated cellulose. The alkali cellulose is then reacted with ethylene oxide to produce a series of hydroxyethyl cellulose ethers. The manner in which ethylene oxide is added to cellulose can be described by two terms, the degree of substitution (DS) and the molar substitution (MS). The DS designates the average number of hydroxyl positions on the anhydroglucose unit that have been reacted with ethylene oxide. Since each anhydroglucose unit of the cellulose molecule has three hydroxyl groups, the maximum value for DS is 3. MS is defined as the average number of ethylene oxide molecules that have reacted with each anhydroglucose unit. Once a hydroxyethyl group is attached to each unit, it can further react with additional groups in an end-to-end formation. This reaction can continue and there is no theoretical limit for MS.
Hydroxyethyl cellulose is primarily used in ophthalmic and topical pharmaceutical formulations. It is generally regarded as an essentially nontoxic and nonirritant material.(7,8) Acute and subacute oral toxicity studies in rats have shown no toxic effects attributable to hydroxyethyl cellulose consumption, the hydroxyethyl cellulose being neither absorbed nor hydrolyzed in the rat gastrointestinal tract. However, although used in oral pharmaceutical formulations, hydroxyethyl cellulose has not been approved for direct use in food products; see Section 16. Glyoxal-treated hydroxyethyl cellulose is not recommended for use in oral pharmaceutical formulations or topical preparations that may be used on mucous membranes. Hydroxyethyl cellulose is also not recommended for use in parenteral products.
Observe normal precautions appropriate to the circumstances and quantity of material handled. Hydroxyethyl cellulose dust may be irritant to the eyes, and eye protection is recommended. Excessive dust generation should be avoided to minimize the risks of explosion. Hydroxyethyl cellulose is combustible. When heated to decomposition, hydroxyethyl cellulose emits acrid smoke and irritating vapors, in which case a ventilator is recommended.
Included in the FDA Inactive Ingredients Database (ophthalmic preparations; oral syrups and tablets; otic and topical preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients. Hydroxyethyl cellulose is not currently approved for use in food products in Europe or the USA, although it is permitted for use in indirect applications such as packaging. This restriction is due to the high levels of ethylene glycol residues that are formed during the manufacturing process.
Hydroxyethylmethyl cellulose; hydroxypropyl cellulose; hydroxypropyl cellulose, low-substituted; hypromellose; methylcellulose.
Hydroxyethyl cellulose is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State ofWork’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. The limited scope for the use of hydroxyethyl cellulose in foodstuffs is in stark contrast to its widespread application as an excipient in oral pharmaceutical formulations. Hydroxyethyl cellulose hydrogels may also be used in various delivery systems.(9)