

Name: Methylcellulose
CAS No: Cellulose methyl ether [9004-67-5]
BP: Methylcellulose JP: Methylcellulose PhEur: Methylcellulose USP: Methylcellulose
Benecel; Cellacol; Culminal MC; E461; Mapolose; Methocel; methylcellulosum; Metolose; Tylose; Viscol
Cellulose methyl ether [9004-67-5]
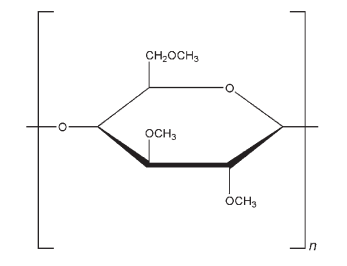
Methylcellulose is a long-chain substituted cellulose in which approximately 27–32% of the hydroxyl groups are in the form of the methyl ether. The various grades of methylcellulose have degrees of polymerization in the range 50–1000, with molecular weights (number average) in the range 10 000–220 000 Da. The degree of substitution of methylcellulose is defined as the average number of methoxyl (CH3O) groups attached to each of the anhydroglucose units along the chain. The degree of substitution also affects the physical properties of methylcellulose, such as its solubility.
The structure shown is with complete substitution of the available hydroxyl units of methoxyl substitution. Note that methoxyl substitution can occur at any combination of the hydroxyl groups of the anhydroglucose ring of cellulose at positions 2, 3, and 6. See Section 4
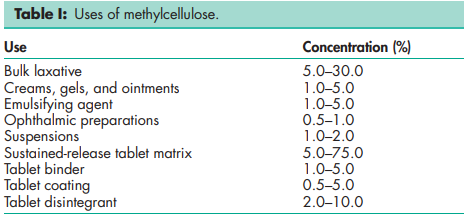
Methylcellulose is widely used in oral and topical pharmaceutical formulations; see Table I. In tablet formulations, low- or medium-viscosity grades of methylcellulose are used as binding agents, the methylcellulose being added either as a dry powder or in solution.(1–3) Highviscosity grades of methylcellulose may also be incorporated in tablet formulations as a disintegrant.(4) Methylcellulose may be added to a tablet formulation to produce sustained-release preparations.(5) Tablet cores may also be spray-coated with either aqueous or organic solutions of highly substituted low-viscosity grades of methylcellulose to mask an unpleasant taste or to modify the release of a drug by controlling the physical nature of the granules.(6) Methylcellulose coats are also used for sealing tablet cores prior to sugar coating. Low-viscosity grades of methylcellulose are used to emulsify olive, peanut, and mineral oils.(7) They are also used as suspending or thickening agents for orally administered liquids, methylcellulose commonly being used in place of sugar-based syrups or other suspension bases.(8) Methylcellulose delays the settling of suspensions and increases the contact time of drugs, such as antacids, in the stomach. High-viscosity grades of methylcellulose are used to thicken topically applied products such as creams and gels. In ophthalmic preparations, a 0.5–1.0% w/v solution of a highly substituted, high-viscosity grade of methylcellulose has been used as a vehicle for eye drops.(9) However, hypromellose-based formulations are now preferred for ophthalmic preparations. Methylcellulose is also used in injectable formulations. Therapeutically, methylcellulose is used as a bulk laxative; it has also been used to aid appetite control in the management of obesity, but there is little evidence supporting its efficacy
Methylcellulose occurs as a white, fibrous powder or granules. It is practically odorless and tasteless. It should be labeled to indicate its viscosity type (viscosity of a 1 in 50 solution).
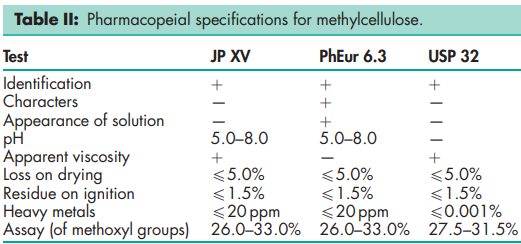
See Table II.
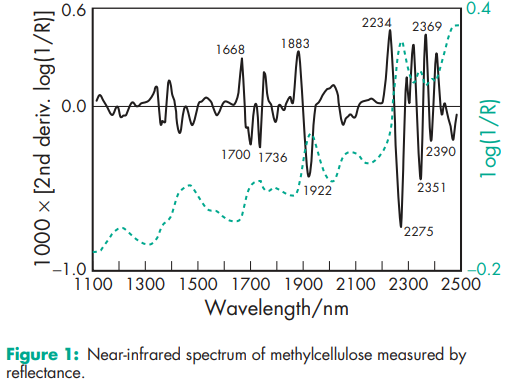
Acidity/alkalinity pH = 5.0–8.0 for a 1% w/v aqueous suspension. Autoignition temperature >3508C for Methocel A4M. Degree of substitution 1.64–1.92 Density (bulk) 0.276 g/cm3 Density (tapped) 0.464 g/cm3 Density (true) 1.341 g/cm3 Glass transition temperature (Tg) 1968C for Methocel A4M. Melting point Begins to brown at 190–2008C; begins to char at 225–2308C. NIR spectra see Figure 1. Refractive index of solution nD 20 = 1.336 (2% aqueous solution). Solubility Practically insoluble in acetone, methanol, chloroform, ethanol (95%), ether, saturated salt solutions, toluene, and hot water. Soluble in glacial acetic acid and in a mixture of equal volumes of ethanol and chloroform. In cold water, methylcellulose swells and disperses slowly to form a clear to opalescent, viscous, colloidal dispersion. Surface tension 53–59 mN/m (53–59 dynes/cm) for a 0.05% w/v solution at 258C; 45–55 mN/m for 0.1% at 208C. Interfacial tension of solution versus paraffin oil is 19–23 mN/m for 0.1% w/v solution at 208C. Viscosity (dynamic) Various grades of methylcellulose are commercially available that vary in their degree of polymerization. Aqueous solutions at concentrations of 2% w/v will produce viscosities between 5 and 75 000 mPa s. Individual grades of methylcellulose have a stated, narrowly defined viscosity range measured for a 2% w/v solution. The viscosity of solutions may be increased by increasing the concentration of methylcellulose. Increased temperatures reduce the viscosity of solutions until gel formation occurs at 50–608C. The process of thermogelation is reversible, with a viscous solution being reformed on cooling. See also Table III.


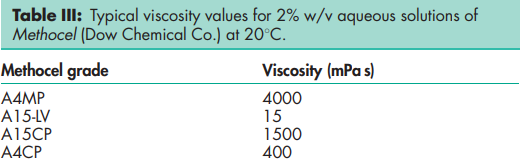
Methylcellulose powder is stable, although slightly hygroscopic. The bulk material should be stored in an airtight container in a cool, dry place. Solutions of methylcellulose are stable to alkalis and dilute acids at pH 3–11, at room temperature. At pH less than 3, acid-catalyzed hydrolysis of the glucose–glucose linkages occurs and the viscosity of methylcellulose solutions is reduced.(10) On heating, solution viscosity is reduced until gel formation occurs at approximately 508C; see Section 10. Methylcellulose solutions are liable to microbial spoilage and antimicrobial preservatives should therefore be used. Solutions may also be sterilized by autoclaving, although this process can decrease the viscosity of a solution.(11,12) The change in viscosity after autoclaving is related to solution pH. Solutions at pH less than 4 had viscosities reduced by more than 20% subsequent to autoclaving.(11
Methylcellulose is incompatible with aminacrine hydrochloride; chlorocresol; mercuric chloride; phenol; resorcinol; tannic acid; silver nitrate; cetylpyridinium chloride; p-hydroxybenzoic acid; paminobenzoic acid; methylparaben; propylparaben; and butylparaben. Salts of mineral acids (particularly polybasic acids), phenols, and tannins will coagulate solutions of methylcellulose, although this can be prevented by the addition of ethanol (95%) or glycol diacetate. Complexation of methylcellulose occurs with highly surface-active compounds such as tetracaine and dibutoline sulfate. High concentrations of electrolytes increase the viscosity of methylcellulose mucilages owing to the ‘salting out’ of methylcellulose. With very high concentrations of electrolytes, the methylcellulose may be completely precipitated in the form of a discrete or continuous gel. Methylcellulose is incompatible with strong oxidizing agents.
Methylcellulose is prepared from wood pulp (cellulose) by treatment with alkali followed by methylation of the alkali cellulose with methyl chloride. The product is then purified and ground to powder form.
Methylcellulose is widely used in a variety of oral and topical pharmaceutical formulations. It is also extensively used in cosmetics and food products, and is generally regarded as a nontoxic, nonallergenic, and nonirritant material.(13) Following oral consumption, methylcellulose is not digested or absorbed and is therefore a noncaloric material. Ingestion of excessive amounts of methylcellulose may temporarily increase flatulence and gastrointestinal distension. In the normal individual, oral consumption of large amounts of methylcellulose has a laxative action and medium- or high-viscosity grades are therefore used as bulk laxatives. Esophageal obstruction may occur if methylcellulose is swallowed with an insufficient quantity of liquid. Consumption of large quantities of methylcellulose may additionally interfere with the normal absorption of some minerals. However, this and the other adverse effects discussed above relate mainly to the use of methylcellulose as a bulk laxative and are not significant factors when methylcellulose is used as an excipient in oral preparations. Methylcellulose is not commonly used in parenteral products, although it has been used in intra-articular and intramuscular injections. Studies in rats have suggested that parenterally administered methylcellulose may cause glomerulonephritis and hypertension.(13) Methylcellulose is considered to be toxic by the intraperitoneal route of administration. The WHO has not specified an acceptable daily intake of methylcellulose since the level of use in foods was not considered to be a hazard to health.(14) LD50 (mouse, IP): 275 g/kg(15)
Observe normal precautions appropriate to the circumstances and quantity of material handled. Dust may be irritant to the eyes and eye protection should be worn. Use in a well-ventilated area. Excessive dust generation should be avoided to minimize the risk of explosion. Methylcellulose is combustible. Spills of the dry powder or solution should be cleaned up immediately, as the slippery film that forms can be dangerous.
GRAS listed. Accepted as a food additive in the USA, Europe and Japan. Included in the FDA Inactive Ingredients Database (sublingual tablets; IM injections; intrasynovial injections; nasal preparations; ophthalmic preparations; oral capsules, oral suspensions, and oral tablets; topical and vaginal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.Reported in the EPA TSCA inventory
Ethylcellulose; hydroxyethyl cellulose; hydroxyethylmethyl cellulose; hypromellose
Methylcellulose is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. The thermal gelation temperature for methylcellulose decreases as a function of concentration. The presence of additives can increase or decrease the thermal gelation temperature. The presence of drugs can influence the properties of methylcellulose gels.(16) In addition, the viscosity of methylcellulose solutions can be modified by the presence of drugs or other additives.(17) Aqueous solutions of methylcellulose can be frozen and do not undergo phase separation upon freezing. Methylcellulose is best dissolved in water by one of three methods, the most suitable being chosen for a particular application. The most commonly used method is to add methylcellulose initially to hot water. The appropriate quantity of methylcellulose required to produce a solution of specified viscosity is mixed with water at 708C; about half the desired final volume of water is used. Cold water or ice is then added to the hot methylcellulose slurry in order to reduce the temperature to below 208C. A clear, aqueous methylcellulose solution is obtained. Alternatively, either methylcellulose powder may be dry-blended with another powder prior to mixing with cold water, or methylcellulose powder may be moistened with an organic solvent such as ethanol (95%) prior to the addition of water. In general, methylcellulose solutions exhibit pseudoplastic flow and there is no yield point. Nonthixotropic flow properties are observed below the gelation temperature. bstitutions in addition to methyl substitutions but are designated with the same trade name in a product line, differing only by a unique identifier code. These products should not be confused with the products that contain only methyl substitutions. Methylcellulose has been investigated as a stabilizer for liposome dispersions.(18) A specification for methylcellulose is contained in the Food Chemicals Codex (FCC).(19)