

Name: Wax, Microcrystalline
CAS No: Microcrystalline wax [63231-60-7]
USP-NF: Microcrystalline Wax
Amorphous wax; E907; petroleum ceresin; petroleum wax (microcrystalline).
Microcrystalline wax [63231-60-7]
Microcrystalline wax is composed of a mixture of straight-chain and randomly branched saturated alkanes obtained from petroleum. The carbon chain lengths range from C41 to C57; cyclic hydrocarbons are also present.
Microcrystalline wax is used mainly as a stiffening agent in topical creams and ointments. The wax is used to modify the crystal structure of other waxes (particularly paraffin wax) present in a mixture so that changes incrystal structure, usually exhibited over a period of time, do not occur. Microcrystalline wax also minimizes the sweating or bleeding of oils from blends of oils and waxes. Microcrystalline wax generally has a higher melting point than paraffin wax, and higher viscosity when molten, thereby increasing the consistency of creams and ointments when incorporated into such formulations. Microcrystalline wax is also used in oral controlled-release matrix pellet formulations for various active compounds(1–3) and as a tablet- and capsule-coating agent. In controlled-release systems, microcrystalline wax coatings can also be used to affect the release of drug from ion-exchange resin beads.(4) Microcrystalline wax is also used in confectionery, cosmetics, and food products.
Microcrystalline wax occurs as odorless and tasteless waxy lumps or flakes containing small irregularly shaped crystals. It may vary in color from white to yellow, amber, brown, or black depending on the grade of material; pharmaceutical grades are usually white or yellow. The USP32–NF27 describes microcrystalline wax as a mixture of straight-chain, branched-chain, and cyclic hydrocarbons, obtained by solvent fractionation of the still-bottom fraction of petroleum by suitable means of dewaxing or de-oiling.
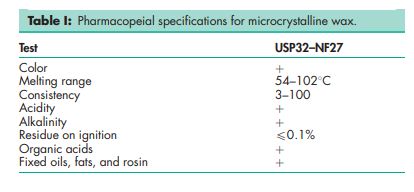
See Table I.
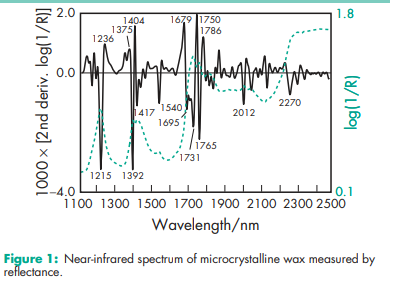
Acid value 1.0 Density 0.928–0.941 g/cm3 Freezing point 60.0–75.08C NIR spectra see Figure 1. Refractive index nD 100 = 1.435–1.445 Saponification value 0.05–0.10 Solubility Soluble in benzene, chloroform, and ether; slightly soluble in ethanol; practically insoluble in water. When melted, microcrystalline wax is miscible with volatile oils and most warm fixed oils. Viscosity (dynamic) 10.0–30.0 mPa s (10.0–30.0 cP) at 1008C.
Microcrystalline wax is stable in the presence of acids, alkalis, light, and air. The bulk material should be stored in a well-closed container in a cool, dry place.
—
Microcrystalline wax is obtained by solvent fractionation of the still-bottom fraction of petroleum by suitable dewaxing or deoiling.
Microcrystalline wax is mainly used in topical pharmaceutical formulations but is also used in some oral products. It is generally regarded as a nontoxic and nonirritating material.
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection is recommended.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (oral capsules; topical and vaginal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Paraffin; wax, white; wax, yellow.
Rheological studies of a model ointment containing microcrystalline wax, white petroleum, and mineral oil showed that while the latter two substances control the rheology of the ointment, microcrystalline wax incorporates itself into the existing white petroleum structure and builds up the structure of the ointment.(5)