

Name: Benzyl Benzoate
CAS No: Benzoic acid phenylmethyl ester [120-51-4]
BP: Benzyl Benzoate JP: Benzyl Benzoate PhEur: Benzyl Benzoate USP: Benzyl Benzoate
Benzoic acid benzyl ester; benzylbenzenecarboxylate; benzylis benzoas; benzyl phenylformate; phenylmethyl benzoate.
Benzoic acid phenylmethyl ester [120-51-4]
C14H12O2 212.24
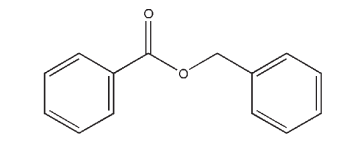
Benzyl benzoate is used as a solubilizing agent and nonaqueous solvent in intramuscular injections at concentrations of 0.01–46.0% v/v,(1) and as a solvent and plasticizer for cellulose and nitrocellulose. It is also used in the preparation of spray-dried powders using nanocapsules.(2) However, the most widespread pharmaceutical use of benzyl benzoate is as a topical therapeutic agent in the treatment of scabies.(3) Benzyl benzoate is also used therapeutically as a parasiticide in veterinary medicine.(4) Other applications of benzyl benzoate include its use as a pediculicide, and as a solvent and fixative for flavors and perfumes in cosmetics and food products.
Benzyl benzoate is a clear, colorless, oily liquid with a slightly aromatic odor. It produces a sharp, burning sensation on the tongue. At temperatures below 178C it exists as clear, colorless crystals.
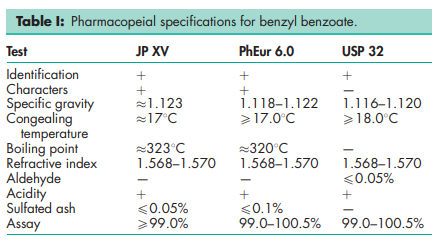
See Table I
Autoignition temperature 4818C Boiling point 323–3248C Flash point 1488C Freezing point 178C Partition coefficient Octanol : water log kow = 3.97 Refractive index n D 21 = 1.5681 Solubility Soluble in acetone and benzene; practically insoluble in glycerin and water; miscible with chloroform, ethanol (95%), ether, and with fatty acids and essential oils. Specific gravity 1.12 Vapor density (relative) 7.3 (air = 1)
Benzyl benzoate is stable when stored in tight, well-filled, lightresistant containers. Exposure to excessive heat (above 408C) should be avoided.
Benzyl benzoate is incompatible with alkalis and oxidizing agents
Benzyl benzoate is a constituent of Peru balsam and occurs naturally in certain plant species. Commercially, benzyl benzoate is produced synthetically by the dry esterification of sodium benzoate and benzoyl chloride in the presence of triethylamine or by the reaction of sodium benzylate with benzaldehyde.
Benzyl benzoate is metabolized by rapid hydrolysis to benzoic acid and benzyl alcohol. Benzyl alcohol is then further metabolized to hippuric acid, which is excreted in the urine. Benzyl benzoate is widely used as a 25% v/v topical application in the treatment of scabies and as an excipient in intramuscular injections and oral products. Adverse reactions to benzyl benzoate include skin irritation and hypersensitivity reactions. Oral ingestion may cause harmful stimulation of the CNS and convulsions. Benzyl benzoate should be avoided by perople with perfume allergy.(5) LD50 (cat, oral): 2.24 g/kg(6–9) LD50 (dog, oral): 22.44 g/kg LD50 (guinea pig, oral): 1.0 g/kg LD50 (mouse, oral): 1.4 g/kg LD50 (rabbit, oral): 1.68 g/kg LD50 (rabbit, skin): 4.0 g/kg LD50 (rat, oral): 0.5 g/kg LD50 (rat, skin): 4.0 g/kg
Benzyl benzoate may be harmful if ingested, and is irritating to the skin, eyes, and mucous membranes. Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection, gloves, and a respirator are recommended. It is recommended that benzyl benzoate is handled in a fume cupboard. Benzyl benzoate is flammable.
Included in the FDA Inactive Ingredients Database (IM injections and oral capsules). Included, as an active ingredient, in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Benzyl benzoate has been shown to have an inhibitory effect on angiotensin II-induced hypertension.(10) The EINECS number for benzyl benzoate is 204-402-9. The PubChem Compound ID (CID) for benzyl benzoate is 2345.