

Name: Monoethanolamine
CAS No: 2-Aminoethanol [141-43-5]
BP: Ethanolamine USP-NF: Monoethanolamine
b-Aminoethyl alcohol; colamine; ethylolamine; b-hydroxyethylamine; 2-hydroxyethylamine.
2-Aminoethanol [141-43-5]
C2H7NO 61.08
Monoethanolamine is used primarily in pharmaceutical formulations for buffering purposes and in the preparation of emulsions. Other uses include as a solvent for fats and oils and as a stabilizing agent in an injectable dextrose solution of phenytoin sodium. Monoethanolamine is also used to produce a variety of salts with therapeutic uses. For example, a salt of monoethanolamine with vitamin C is used for intramuscular injection, while the salicylate and undecenoate monoethanolamine salts are utilized respectively in the treatment of rheumatism and as an antifungal agent. However, the most common therapeutic use of monoethanolamine is in the production of ethanolamine oleate injection, which is used as a sclerosing agent.(1)
Monoethanolamine is a clear, colorless or pale yellow-colored, moderately viscous liquid with a mild, ammoniacal odor.
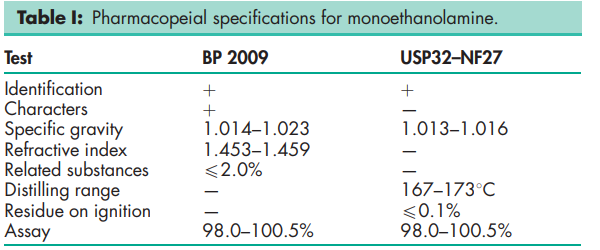
See Table I.
Acidity/alkalinity pH = 12.1 for a 0.1 N aqueous solution. Boiling point 170.88C Critical temperature 3418C Density 1.0117 g/cm3 at 258C; 0.9998 g/cm3 at 408C; 0.9844 g/cm3 at 608C. Dissociation constant pKa = 9.4 at 258C Flash point (open cup) 938C Hygroscopicity Very hygroscopic. Melting point 10.38C Refractive index nD 20 = 1.4539 Solubility Surface tension 48.8 mN/m at 208C Vapor density (relative) 2.1 (air = 1) Vapor pressure 53.3 Pa (0.4 mmHg) at 208C Viscosity (dynamic) 18.95 mPa s (18.95 cP) at 258C; 5.03 mPa s (5.03 cP) at 608C.

Monoethanolamine is very hygroscopic and is unstable when exposed to light. Aqueous monoethanolamine solutions may be sterilized by autoclaving. When monoethanolamine is stored in large quantities, stainless steel is preferable for long-term storage. Copper, copper alloys, zinc, and galvanized iron are corroded by amines and should not be used for construction of storage containers. Ethanolamines readily absorb moisture and carbon dioxide from the air; they also react with carbon dioxide. This can be prevented by sealing the monoethanolamine under an inert gas. Smaller quantities of monoethanolamine should be stored in an airtight container, protected from light, in a cool, dry place.
Monoethanolamine contains both a hydroxy group and a primary amine group and will thus undergo reactions characteristic of both alcohols and amines. Ethanolamines will react with acids to form salts and esters. Discoloration and precipitation will take place in the presence of salts of heavy metals. Monoethanolamine reacts with acids, acid anhydrides, acid chlorides, and esters to form amide derivatives, and with propylene carbonate or other cyclic carbonates to give the corresponding carbonates. As a primary amine, monoethanolamine will react with aldehydes and ketones to yield aldimines and ketimines. Additionally, monoethanolamine will react with aluminum, copper, and copper alloys to form complex salts. A violent reaction will occur with acrolein, acrylonitrile, epichlorohydrin, propiolactone, and vinyl acetate.
Monoethanolamine is prepared commercially by the ammonolysis of ethylene oxide. The reaction yields a mixture of monoethanolamine, diethanolamine, and triethanolamine, which is separated to obtain the pure products. Monoethanolamine is also produced from the reaction between nitromethane and formaldehyde.
Monoethanolamine is an irritant, caustic material, but when it is used in neutralized parenteral and topical pharmaceutical formulations it is not usually associated with adverse effects, although hypersensitivity reactions have been reported. Monoethanolamine salts are generally regarded as being less toxic than monoethanolamine. LD50 (mouse, IP): 0.05 g/kg(2) LD50 (mouse, oral): 0.7 g/kg LD50 (rabbit, skin): 1.0 g/kg LD50 (rat, IM): 1.75 g/kg LD50 (rat, IP): 0.07 g/kg LD50 (rat, IV): 0.23 g/kg LD50 (rat, oral): 1.72 g/kg LD50 (rat, SC): 1.5 g/kg
When handling concentrated solutions of monoethanolamine, personal protective equipment such as an appropriate respirator, chemically resistant gloves, safety goggles, and other protective clothing should be worn. Transfer or prepare monoethanolamine solutions only in a chemical fume hood. Vapors may flow along surfaces to distant ignition sources and flash back. Closed containers exposed to heat may explode. Contact with strong oxidizers may cause fire. In the UK, the short-term (15-minute) workplace exposure limit for monoethanolamine is 7.6 mg/m3 (3 ppm) and the long-term exposure limit (8-hour TWA) is 2.5 mg/m3 (1 ppm).(3
Included in parenteral and nonparenteral medicines licensed in the UK and USA. Included in the Canadian List of Acceptable Nonmedicinal Ingredients.
Diethanolamine; triethanolamine
The EINECS number for monoethanolamine is 205-483-3.