

Name: Wax, White
CAS No: White beeswax [8012-89-3]
BP: White Beeswax JP: White Beeswax PhEur: Beeswax, White USP-NF: White Wax
Bleached wax; cera alba; E901.
White beeswax [8012-89-3]
White wax is the chemically bleached form of natural beeswax; see Section 13. Beeswax consists of 70–75% of a mixture of various esters of straight-chain monohydric alcohols with even-numbered carbon chains from C24 to C36 esterified with straight-chain acids. These straight-chain acids also have even numbers of carbon atoms up to C36 together with some C18 hydroxy acids. The chief ester is myricyl palmitate. Also present are free acids (about 14%) and carbohydrates (about 12%) as well as approximately 1% free wax alcohols and stearic esters of fatty acids.
White wax is a chemically bleached form of yellow wax and is used in similar applications: for example, to increase the consistency of creams and ointments, and to stabilize water-in-oil emulsions. White wax is used to polish sugar-coated tablets and to adjust the melting point of suppositories. White wax is also used as a film coating in sustained-release tablets.(1) White beeswax microspheres may be used in oral dosage forms to retard the absorption of an active ingredient from the stomach, allowing the majority of absorption to occur in the intestinal tract. Wax coatings can also be used to affect the release of drug from ion-exchange resin beads.(2–4)
White wax consists of tasteless, white or slightly yellow-colored sheets or fine granules with some translucence. Its odor is similar to that of yellow wax but is less intense.
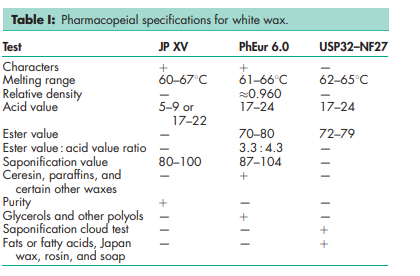
See Table I.
Arsenic 43 ppm Density 0.95–0.96 g/cm3 Flash point 245–2588C Heavy metals 40.004% Iodine number 8–11 Lead 410 ppm Melting point 61–658C NIR spectra see Figure 1. Peroxide value 48 Solubility Soluble in chloroform, ether, fixed oils, volatile oils, and warm carbon disulfide; sparingly soluble in ethanol (95%); practically insoluble in water. Unsaponified matter 52–55%
When the wax is heated above 1508C, esterification occurs with a consequent lowering of acid value and elevation of melting point. White wax is stable when stored in a well-closed container, protected from light
Incompatible with oxidizing agents.
Yellow wax (beeswax) is obtained from the honeycomb of the bee (Apis mellifera Linne´ (Fam. Apidae)); see Wax, Yellow. Subsequent treatment with oxidizing agents bleaches the wax to yield white wax.
White wax is used in both topical and oral formulations, and is generally regarded as an essentially nontoxic and nonirritant material. However, although rare, hypersensitivity reactions to beeswax (attributed to contaminants in the wax) have been reported.(5,6)
Observe normal precautions appropriate to the circumstances and quantity of material handled.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (oral capsules and tablets; rectal, topical, and vaginal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Paraffin; wax, microcrystalline; wax, yellow.
—