

Name: Myristyl Alcohol
CAS No: Tetradecan-1-ol [112-72-1]
USP-NF: Myristyl Alcohol
Alcohol miristilo; Dytol R-52; Lanette Wax KS; Lorol C14-95; Loxanol V; myristic alcohol; Nacol 14-95; Nacol 14-98; 1- tetradecanol; n-tetradecanol-1; n-tetradecyl alcohol; tetradecyl alcohol; Unihydag WAX-14.
Tetradecan-1-ol [112-72-1]
C14H30O 214.4
Myristyl alcohol is used in oral, parenteral, and topical pharmaceutical formulations. It has been evaluated as a penetration enhancer in melatonin transdermal patches in rats.(1) Myristyl alcohol has also been tested as a bilayer stabilizer in niosome formulations containing ketorolac tromethamine,(2) and zidovudine.(3) Niosomes containing myristyl alcohol showed a considerably slower release rate of ketorolac tromethamine than those containing cholesterol.(2) This was also observed with the zidovudine formulation.(3
Myristyl alcohol occurs as a white crystalline solid with a waxy odor. Also reported as opaque leaflets or crystals from ethanol.(4)
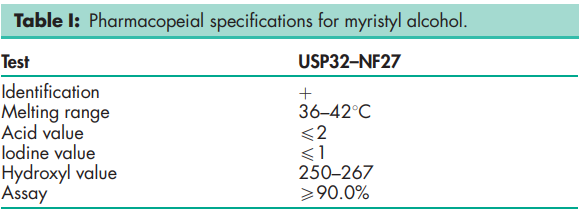
See Table I.
Boiling point 1678C at 1.5 mPa (15 atm) Density 0.8355 g/cm3 at 208C for solid; 0.8236 g/cm3 at 388C for liquid(4) Flash point 1408C (open cup)(4) Melting point 388C; also reported as 37.68C(4) Solubility Practically insoluble in water; soluble in ether, slightly soluble in ethanol (95%) Specific gravity 0.824 Vapor pressure 1.33 Pa (0.01 mmHg) at 208C(4)
The bulk material should be stored in a well-closed container in a cool, dry place.
Myristyl alcohol is combustible when exposed to heat or flame. It can react with oxidizing materials. When heated to decomposition, it emits acrid smoke and irritating fumes.(4)
Myristyl alcohol is found in spermaceti wax and sperm oil, and may be synthesized by sodium reduction of fatty acid esters or the reduction of fatty acids by lithium aluminum hydride. It can also be formed from acetaldehyde and dimethylamine.(5
Myristyl alcohol is used in oral parenteral, and topical pharmaceutical formulations. The pure form of myristyl alcohol is mildly toxic by ingestion and may be carcinogenic; experimental tumorigenic data are available.(4) It is also a human skin irritant. In animal studies of the skin permeation enhancement effect of saturated fatty alcohols, myristyl alcohol exhibited a lower effect when compared with decanol, undecanol, or lauryl alcohol but caused greater skin irritation.(6)A study investigating contact sensitization to myristyl alcohol revealed that patch testing of myristyl alcohol 10% petrolatum should not be carried out owing to observed irritant effects; thus the use of a lower concentration of myristyl alcohol for such tests (5% petrolatum) was recommended.(7) Myristyl alcohol has been associated with some reports of contact allergy.(8,9)A moderate-to-severe erythema and moderate edema are seen when 75 mg is applied to human skin intermittently in three doses over 72 hours.(4) LD50(rabbit, skin): 7.1 g/kg(4) LD50(rat, oral): 33.0 g/kg(4
Observe normal precautions appropriate to the circumstances and quantity of the material handled. The use of gloves is recommended.
Included in the FDA Inactive Ingredients Database (oral tablet: sustained-release; and topical formulations: cream, lotion, suspension). Included in nonparenteral (topical cream) formulations licensed in the UK.
Lauric acid; myristic acid; palmitic acid; potassium myristate; sodium myristate; stearic acid.
The steady-state flux value of melatonin across human skin using myristyl alcohol as a permeation enhancer was reported as 18.2 mg/ (cm3 h).(10) The EINECS number for myristyl alcohol is 204-000-3. The PubChem Compound ID (CID) for myristyl alcohol is 8209