

Name: Boric Acid
CAS No: Orthoboric acid [10043-35-3], Metaboric acid [13460-50-9]
BP: Boric Acid JP: Boric Acid PhEur: Boric Acid USP-NF: Boric Acid
Acidum boricum; boracic acid; boraic acid; Borofax; boron trihydroxide; E284; orthoboric acid; trihydroxyborene
Orthoboric acid [10043-35-3], Metaboric acid [13460-50-9]
H3BO3 61.83 (for trihydrate) HBO2 43.82 (for monohydrate)
See Section 4.
Boric acid is used as an antimicrobial preservative(1) in eye drops, cosmetic products, ointments, and topical creams. It is also used as an antimicrobial preservative in foods. Boric acid and borate have good buffering capacity and are used to control pH; they have been used for this purpose in external preparations such as eye drops.(2) Boric acid has also been used therapeutically in the form of suppositories to treat yeast infections.(3,4) In dilute concentrations it is used as a mild antiseptic, with weak bacteriostatic and fungistatic properties, although it has generally been superseded by more effective and less toxic disinfectants.(5) See Section 14.
Boric acid occurs as a hygroscopic, white crystalline powder, colorless shiny plates, or white crystals.
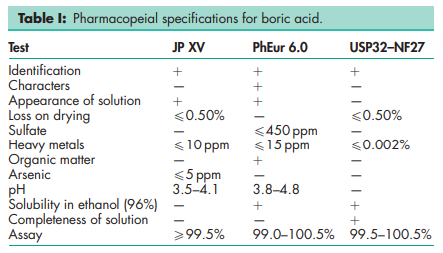
See Table I
Acidity/alkalinity pH = 3.5–4.1 (5% w/v aqueous solution) Density 1.435 Melting point 170.98C. When heated slowly to 181.08C, boric acid loses water to form metaboric acid (HBO2); tetraboric acid (H2B4O7) and boron trioxide (B2O3) are formed at higher temperatures.(6) Solubility Soluble in ethanol, ether, glycerin, water, and other fixed and volatile oils. Solubility in water is increased by addition of hydrochloric, citric, or tartaric acids. Specific gravity 1.517
Boric acid is hygroscopic and should therefore be stored in an airtight, sealed container. The container must be labeled ‘Not for Internal Use’. SEM 1: Excipient: boric acid; manufacturer: Alfa Aesar; lot no.: 23672; magnification: 100; voltage: 5 kV. SEM 2: Excipient: boric acid; manufacturer: Aldrich Chemical Company Inc.; lot no.: 01559BU; magnification: 100; voltage: 5 kV.


Boric acid is incompatible with water, strong bases and alkali metals. It reacts violently with potassium and acid anhydrides. It also forms a complex with glycerin, which is a stronger acid than boric acid
Boric acid occurs naturally as the mineral sassolite. However, the majority of boric acid is produced by reacting inorganic borates with sulfuric acid in an aqueous medium. Sodium borate and partially refined calcium borate (colemanite) are the principal raw materials. When boric acid is made from colemanite, the fineground ore is vigorously stirred with mother liquor and sulfuric acid at about 908C. The by-product calcium sulfate is removed by filtration, and the boric acid is crystallized by cooling the filtrate
Boric acid is a weak bacteriostatic and antimicrobial agent, and has been used in topical preparations such as eye lotions, mouthwashes and gargles. It has also been used in US- and Japanese-approved intravenous products. Solutions of boric acid were formerly used to wash out body cavities, and as applications to wounds and ulcers, although the use of boric acid for these purposes is now regarded as inadvisable owing to the possibility of absorption.(5) Boric acid is not used internally owing to its toxicity. It is poisonous by ingestion and moderately toxic by skin contact. Experimentally it has proved to be toxic by inhalation and subcutaneous routes, and moderately toxic by intraperitoneal and intravenous routes. Boric acid is absorbed from the gastrointestinal tract and from damaged skin, wounds, and mucous membranes, although it does not readily permeate intact skin. The main symptoms of boric acid poisoning are abdominal pain, diarrhea, erythematous rash involving both skin and mucous membrane, and vomiting. These symptoms may be followed by desquamation, and stimulation or depression of the central nervous system. Convulsions, hyperpyrexia, and renal tubular damage have been known to occur.(7) Death has occurred from ingestion of less than 5 g in young children, and of 5–20 g in adults. Fatalities have occurred most frequently in young children after the accidental ingestion of solutions of boric acid, or after the application of boric acid powder to abraded skin. The permissible exposure limit (PEL) of boric acid is 15 mg/m3 total dust, and 5 mg/m3 respirable fraction for nuisance dusts.(8) LdLo (man, oral): 429 mg/kg(9) LdLo (woman, oral): 200 mg/kg(9) LdLo (infant, oral): 934 mg/kg(9) LdLo (man, skin): 2.43 g/kg(9) LdLo (infant, skin): 1.20 g/kg(9) LD50 (mouse, oral): 3.45 g/kg(9) LD50 (mouse, IV): 1.24 g/kg LD50 (mouse, SC): 1.74 g/kg LD50 (rat, oral): 2.660 g/kg LD50 (rat, IV): 1.33 g/kg LD50 (rat, SC): 1.4 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Boric acid is irritating to the skin and is potentially toxic by inhalation. Gloves, eye protection, protective clothing, and a respirator are recommended.
Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (IV injections; ophthalmic preparations; (auricular) otic solutions; topical preparations). Reported in the EPA TSCA Inventory. In the UK, the use of boric acid in cosmetics and toiletries is restricted. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Sodium borate.
Boric acid has been used experimentally as a model oxo-acid to retard mannitol crystallization in the solid state.(10) The EINECS number for boric acid is 233-139-2. The PubChem Compound ID (CID) for boric acid includes 7628 and 24492.