Name: Hydroxypropyl Starch
CAS No: Hydroxypropyl starch [113894-92-1]
None adopted.
E1440; hydroxylpropyl starch.
Hydroxypropyl starch [113894-92-1]
Hydroxypropyl starch is a derivative of natural starch; it is described in the JPE 2004 as a hydroxypropyl ether of corn starch.
Hydroxypropyl starch is a modified starch and has been used in combination with carrageenan in the production of soft capsules.(1) Hydroxypropyl starch has been used experimentally in hydrophilic matrices, where it was shown to be an effective matrix for tablets designed for controlled-release drug delivery systems.(2) It has also been used experimentally in the production of hydrophilic matrices by direct compression.(3) It is used in antiseptics and is used widely in cosmetics. It is also used analytically as a bioseparation aqueous-phase-forming polymer.(4)
Hydroxypropyl starch occurs as a free-flowing white to off-white coarse powder.
Acidity/alkalinity pH = 4.5–7.0 (10% w/v aqueous dispersion) Solubility Practically insoluble in water, ethanol (95%), and ether.
Hydroxypropyl starch is stable at high humidity and is considered to be inert under normal conditions. It is stable in emulsion systems at pH 3–9.
Hydroxypropyl starch is produced industrially from natural starch, using propylene oxide as the modifying reagent in the presence of alkali, adding hydroxypropyl (CH(OH)CH2CH3) groups at the OH positions by an ether linkage.
Hydroxypropyl starch is widely used in cosmetics and food products. It is also used in oral pharmaceutical formulations. The WHO has set an acceptable daily intake for hydroxypropyl starch at ‘not limited’ since it was well tolerated on oral consumption.(5) LD50 (rat, oral): 0.218 g/kg(6)
Observe normal precautions appropriate to the circumstances and quantity of material handled.
GRAS listed. Accepted for use as a food additive in Europe.
—
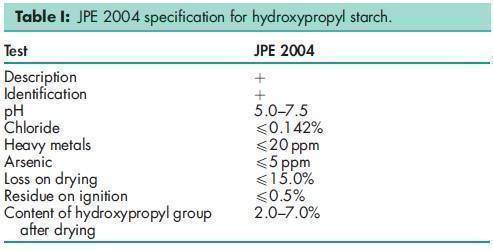
Hydroxypropyl starch–methyl methacrylate (HS-MMA) has been used experimentally in hydrophilic matrices produced by direct compression.(4) Pregelatinized hydroxypropyl starch has been shown to exhibit good disintegrating properties, and can be used as a binder in wet granulation.(7) Although it is not currently included in the pharmacopeias, a specification for hydroxypropyl starch is included in the Japanese Pharmaceutical Excipients (JPE); see Table I.(8) Hydroxypropyl starch is compatible with cationic ingredients (monovalent, divalent), oils, emollients, and silicone. The EINECS number for hydroxypropyl starch is 232-679-6. The PubChem Compound ID (CID) for hydroxypropyl starch is 24847857.