

Name: Hypromellose
CAS No: Cellulose hydroxypropyl methyl ether [9004-65-3]
BP: Hypromellose JP: Hypromellose PhEur: Hypromellose USP: Hypromellose
Benecel MHPC; E464; hydroxypropyl methylcellulose; HPMC; hypromellosum; Methocel; methylcellulose propylene glycol ether; methyl hydroxypropylcellulose; Metolose; MHPC; Pharmacoat; Tylopur; Tylose MO.
Cellulose hydroxypropyl methyl ether [9004-65-3]
The PhEur 6.3 describes hypromellose as a partlyO-methylated and O-(2-hydroxypropylated) cellulose. It is available in several grades that vary in viscosity and extent of substitution. Grades may be distinguished by appending a number indicative of the apparent viscosity, in mPa s, of a 2% w/w aqueous solution at 208C. Hypromellose defined in the USP 32 specifies the substitution type by appending a four-digit number to the nonproprietary name: e.g. hypromellose 1828. The first two digits refer to the approximate percentage content of the methoxy group (OCH3). The second two digits refer to the approximate percentage content of the hydroxypropoxy group (OCH2CH(OH)CH3), calculated on a dried basis. It contains methoxy and hydroxypropoxy groups conforming to the limits for the various types of hypromellose; Molecular weight is approximately 10 000–1 500 000.
Hypromellose is widely used in oral, ophthalmic, nasal, and topical pharmaceutical formulations. In oral products, hypromellose is primarily used as a tablet binder,(1) in film-coating,(2–7) and as a matrix for use in extended release tablet formulations.(8–12) Concentrations between 2% and 5% w/w may be used as a binder in either wet- or dry-granulation processes. High-viscosity grades may be used to retard the release of drugs from a matrix at levels of 10–80% w/w in tablets and capsules. Hypromellose is also used in liquid oral dosage forms as a suspending and/or thickening agent at concentrations ranging from 0.25–5.0%.(13) Depending upon the viscosity grade, concentrations of 2–20% w/w are used for film-forming solutions to film-coat tablets. Lower viscosity grades are used in aqueous film-coating solutions, while higher-viscosity grades are used with organic solvents. Examples of film-coating materials that are commercially available include AnyCoat C, Spectracel, Pharmacoat, and the Methocel E Premium LV series. Hypromellose is also used as a suspending and thickening agent in topical formulations. Compared with methylcellulose, hypromellose produces aqueous solutions of greater clarity, with fewer undissolved fibers present, and is therefore preferred in formulations for ophthalmic use. Hypromellose at concentrations between 0.45–1.0% w/w may be added as a thickening agent to vehicles for eye drops and artificial tear solutions. It is also used commercially in liquid nasal formulations at a concentration of 0.1%.(13) Hypromellose is used as an emulsifier, suspending agent, and stabilizing agent in topical gels and ointments. As a protective colloid, it can prevent droplets and particles from coalescing or agglomerating, thus inhibiting the formation of sediments. In addition, hypromellose is used in the manufacture of capsules, as an adhesive in plastic bandages, and as a wetting agent for hard contact lenses. It is also widely used in cosmetics and food products.
Hypromellose is an odorless and tasteless, white or creamy-white fibrous or granular powder.


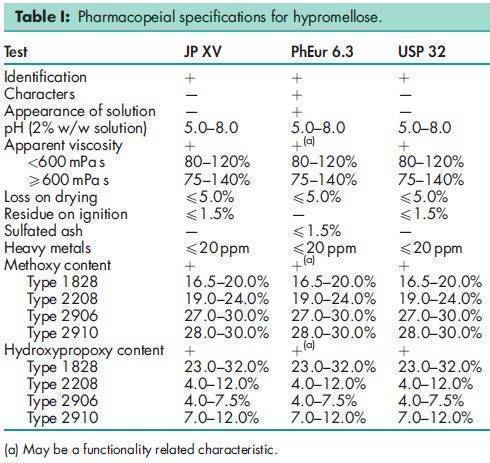
See Table I.
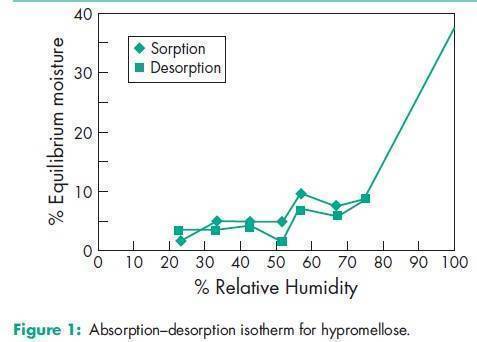
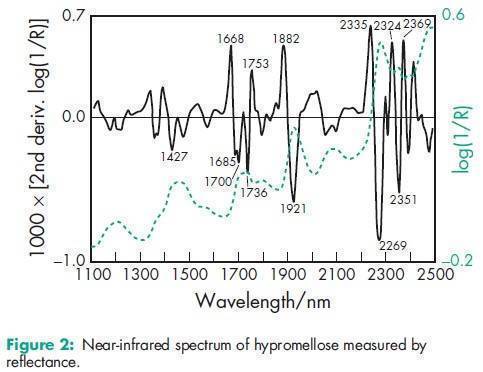
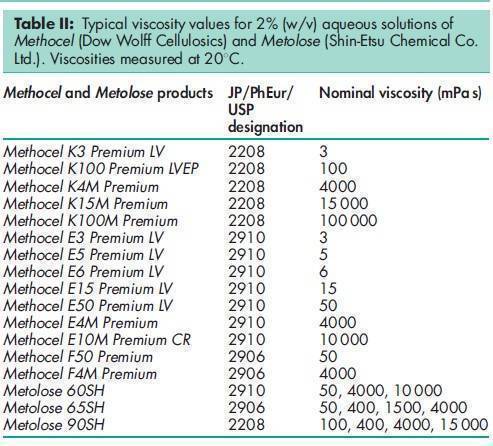
Acidity/alkalinity pH = 5.0–8.0 for a 2% w/w aqueous solution. Ash <_1.5% Autoignition temperature 3608C Density (bulk) 0.341 g/cm3 Density (tapped) 0.557 g/cm3 Density (true) 1.326 g/cm3 Melting point Browns at 190–2008C; chars at 225–2308C. Glass transition temperature is 170–1808C. Moisture content Hypromellose absorbs moisture from the atmosphere; the amount of water absorbed depends upon the initial moisture content and the temperature and relative humidity of the surrounding air. See Figure 1. NIR spectra see Figure 2. Solubility Soluble in cold water, forming a viscous colloidal solution; practically insoluble in hot water, chloroform, ethanol (95%), and ether, but soluble in mixtures of ethanol and dichloromethane, mixtures of methanol and dichloromethane, and mixtures of water and alcohol. Certain grades of hypromellose are soluble in aqueous acetone solutions, mixtures of dichloromethane and propan-2-ol, and other organic solvents. Some grades are swellable in ethanol.(14) See also Section 11. Specific gravity 1.26 Viscosity (dynamic) A wide range of viscosity types are commercially available. Aqueous solutions are most commonly prepared, although hypromellose may also be dissolved in aqueous alcohols such as ethanol and propan-2-ol provided the alcohol content is less than 50% w/w. Dichloromethane and ethanol mixtures may also be used to prepare viscous hypromellose solutions. Solutions prepared using organic solvents tend to be more viscous; increasing concentration also produces more viscous solutions; see Table II. To prepare an aqueous solution, it is recommended that hypromellose is dispersed and thoroughly hydrated in about 20–30% of the required amount of water. The water should be vigorously stirred and heated to 80–908C, and then the hypromellose should be added. The heat source can be removed once the hypromellose has been thoroughly dispersed into the hot water. Sufficient cold water should then be added to produce the required volume while continuing to stir. When aqueous/organic cosolvent mixtures are used for solution preparation, hypromellose should first be dispersed into the organic solvent at a ratio of 5–8 parts of solvent to 1 part of hypromellose. Cold water is then added to produce the final volume. Examples of suitable water-miscible organic solvents include ethanol and glycols. A similar preparation procedure should be used when ethanol and dichloromethane constitute a completely organic cosolvent mixture.
Hypromellose powder is a stable material, although it is hygroscopic after drying. Solutions are stable at pH 3–11. Hypromellose undergoes a reversible sol–gel transformation upon heating and cooling, respectively. The gelation temperature is 50–908C, depending upon the grade and concentration of material. For temperatures below the gelation temperature, viscosity of the solution decreases as temperature is increased. Beyond the gelation temperature, viscosity increases as temperature is increased. Aqueous solutions are comparatively enzyme-resistant, providing good viscosity stability during long-term storage.(15) However, aqueous solutions are liable to microbial spoilage and should be preserved with an antimicrobial preservative: when hypromellose is used as a viscosity-increasing agent in ophthalmic solutions, benzalkonium chloride is commonly used as the preservative. Aqueous solutions may also be sterilized by autoclaving; the coagulated polymer must be redispersed on cooling by shaking. Hypromellose powder should be stored in a well-closed container, in a cool, dry place.
Hypromellose is incompatible with some oxidizing agents. Since it is nonionic, hypromellose will not complex with metallic salts or ionic organics to form insoluble precipitates.
A purified form of cellulose, obtained from cotton linters or wood pulp, is reacted with sodium hydroxide solution to produce a swollen alkali cellulose that is chemically more reactive than untreated cellulose. The alkali cellulose is then treated with chloromethane and propylene oxide to produce methyl hydroxypropyl ethers of cellulose. The fibrous reaction product is then purified and ground to a fine, uniform powder or granules. Hypromellose can then be exposed to anhydrous hydrogen chloride to induce depolymerization, thus producing low viscosity grades.
Hypromellose is widely used as an excipient in oral, opthalmic, nasal, and topical pharmaceutical formulations. It is also used extensively in cosmetics and food products. Hypromellose is generally regarded as a nontoxic and nonirritating material, although excessive oral consumption may have a laxative effect.(16) The WHO has not specified an acceptable daily intake for hypromellose since the levels consumed were not considered to represent a hazard to health.(17) In fact, high dosages of hypromellose are being investigated for treating various metabolic syndromes.(18,19) LD50 (mouse, IP): 5 g/kg(20) LD50 (rat, IP): 5.2 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Hypromellose dust may be irritating to the eyes, so eye protection is recommended. Excessive dust generation should be avoided to minimize the risks of explosion. Hypromellose is combustible.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (ophthalmic and nasal preparations; oral capsules, suspensions, syrups, and tablets; topical and vaginal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Ethylcellulose; hydroxyethyl cellulose; hydroxyethylmethyl cellulose; hydroxypropyl cellulose; hypromellose acetate succinate; hypromellose phthalate; methylcellulose.
Hypromellose has been used in pharmaceutical dosage forms produced using hot-melt extrusion.(21) Premix coating formulations which contain hypromellose as a film-forming agent include Opadry (Colorcon) and Advantia Prime Coating Systems (ISP). Methocel K4MP DC and Methocel K100MP DC (Dow Wolff Cellulosics); they have been developed and commercialized to facilitate direct compression of tablets exhibiting modified-release performance. Powdered or granular, surface-treated grades of hypromellose are also available that are dispersible in cold water. These are not recommended for oral use. A specification for hypromellose is contained in the Food Chemicals Codex (FCC).(22) The PubChem Compound ID (CID) for hypromellose is 24832095.