

Name: Safflower Oil
CAS No: Safflower oil [8001-23-8]
BP: Refined Safflower Oil PhEur: Safflower Oil, Refined USP: Safflower Oil
Aceite de alazor; aceite de cartamo; carthami oleum raffinatum; dygminu aliejus, rafinuotas; huile de carthame; safflorolja, raffinerad; safflower oil (unhydrogenated); saflonoljy puhdistettu.
Safflower oil [8001-23-8]
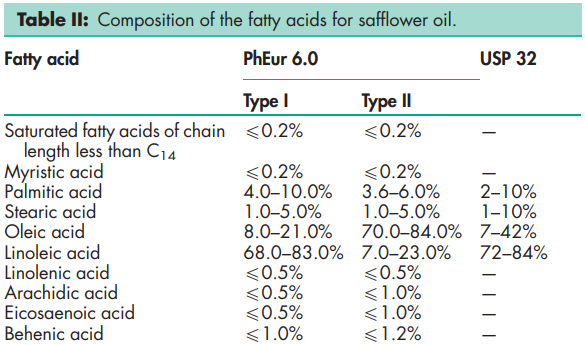
The PhEur 6.0 defines the composition of the fatty acid fraction of two types of refined safflower oil (type I and type II);
See Section 4.
Safflower oil is mainly used as an oleaginous vehicle in oral and topical formulations. It is also used as a component of parenteral fat emulsions for the preparation of parenteral nutrition solutions. Safflower oil has been used as a vehicle in the development of an oral dosage form containing a novel viral-specific inhibitor of the replication of human rhinoviruses.(1) It has also been used as a solvent for a capsule formulation containing a new antilipemic agent; formulations containing safflower oil were found to have the greatest bioavailability in dogs compared with formulations containing PEG 300 or water.(2) A topical lotion containing 3% safflower oil is commercially available, and parenteral fat emulsions containing a mixture of safflower oil 5% and soya oil 5%, or 10% and 10%, respectively, have been administered as part of total parenteral nutrition regimes. Safflower oil is used as a food, being consumed in the form of soft margarine, salad oils, and cooking oils. It is also used in cosmetics products such as soaps, lotions, creams, and hair-care preparations.
Refined safflower oil is a clear, viscous, yellow to pale-yellow liquid, with a slight vegetable odor.
The PhEur 6.0 defines the composition of the fatty acid fraction, refrative index, and relative density values of two types of refined safflower oil (type I and type II);

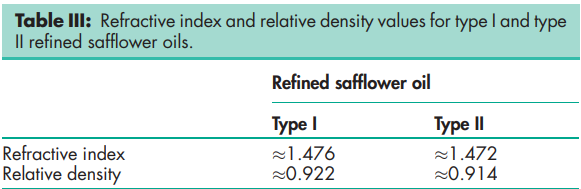
Acid value 1.0–9.7 Flash point >287.88C (closed cup) Hydroxyl value 2.9–6.0 Iodine value 140–150 Refractive index nD 25 = 1.472–1.475; nD 40 = 1.4690–1.4692. See also Table III. Relative density see Table III. Saponification value 188–194 Solubility Soluble in organic solvents. Refined safflower oil is miscible with ether, chloroform, light petroleum (bp 40–608C); practically insoluble in alcohol. Solubility in water is <0.1%
Safflower oil thickens and becomes rancid on prolonged exposure to air. It is also sensitive to light. Safflower oil should be preserved in tight, light-resistant containers. Refined safflower oil should be stored in a well-filled, airtight container, protected from light. Parenteral fat emulsions containing safflower oil are destabilized by electrolytes; severe droplet coalescence in the emulsion occurs 3–5 days after the addition of 10% v/v dimethyl sulfoxide, and after 10 days if 5% v/v is added.(3) Parenteral fat emulsions are prone to bacterial and fungal growth. Generally, fat emulsions containing safflower oil or soybean oil show similar growth patterns,(4,5) although growth of Candida albicans has been reported to be higher in safflower oil containing fat emulsions than in other types of emulsion.(6
Safflower oil is incompatible with strong oxidizing agents.
Refined safflower oil is the fatty oil obtained from the seeds of Carthamus tinctorius L. (type I) or from seeds of hybrids of Carthamus tinctorius L. (type II) by expression and/or extraction followed by refining. Type II refined safflower oil is rich in oleic (cis9-octadecenoic) acid. It may contain a suitable antioxidant. Safflower oil USP 32 is the refined fixed oil yielded by the seed of Carthamus tinctorius Linne´ (Fam. Compositae).
Safflower oil is an edible oil and generally presents no significant health hazards following eye contact, skin contact, oral ingestion, or inhalation. Skin irritation or allergic reactions, or eye irritation may occur. Ingestion of large doses can cause vomiting. Safflower oil may cause diarrhea. LD50 (mouse, IP): >50 g/kg(7)
Observe normal precautions appropriate to the circumstances and quantity of the material handled. When heated to decomposition, safflower oil emits acrid smoke and irritating fumes.
Included in the FDA Inactive Ingredients Database (topical lotion). Included in the Canadian List of Acceptable Non-medicinal Ingredients. Included in an intravenous fat emulsion (Liposyn II) available in the USA. Included in a capsule formulation available in Canada and in a non-medicinal capsule formulation previously available in the UK. It is also a component of a Canadian enteral nutrition preparation.
Almond oil; canola oil; corn oil; cottonseed oil; peanut oil; safflower glycerides; sesame oil; soybean oil; sunflower oil. Safflower glycerides CAS number [79982-97-1] Comments Safflower glycerides (safflower oil monoglycerides) are used in cosmetics as emollients and emulsifying agents. The EINECS number for safflower glycerides is 279-360-8
The PhEur 6.0 requires the label for refined safflower oil to state, where applicable, that the substance is suitable for use in the manufacture of parenteral dosage forms, and the type of oil (type I or type II). In addition, the BP 2009 requires the label to include the name and concentration of any added antioxidant. The PhEur 6.0 also lists a monograph for safflower flower, while JP XV includes an unofficial monograph for safflower. A specification for safflower oil is listed in Japanese Pharmaceutical Excipients (JPE).(8) A monograph for safflower oil (unhydrogenated) is contained in the Food Chemicals Codex (FCC).(9) The EINECS number for safflower oil is 232-276-5.