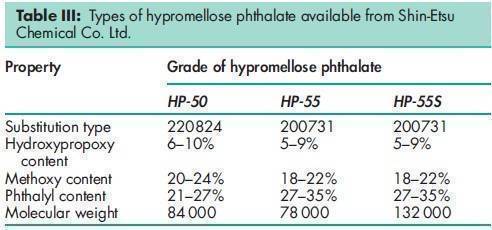
Name: Hypromellose Phthalate
CAS No: Cellulose, hydrogen 1, 2-benzenedicarboxylate, 2-hydroxypropyl methyl ether [9050-31-1]
BP: Hypromellose Phthalate JP: Hypromellose Phthalate PhEur: Hypromellose Phthalate USP-NF: Hypromellose Phthalate
Cellulose phthalate hydroxypropyl methyl ether; HPMCP; hydroxypropyl methylcellulose benzene-1,2-dicarboxylate; 2-hydroxypropyl methylcellulose phthalate; hypromellosi phthalas; Mantrocel HP-55; methylhydroxypropylcellulose phthalate.
Cellulose, hydrogen 1, 2-benzenedicarboxylate, 2-hydroxypropyl methyl ether [9050-31-1]
Hypromellose phthalate is a cellulose in which some of the hydroxyl groups are replaced with methyl ethers, 2-hydroxypropyl ethers, or phthalyl esters. Several different types of hypromellose phthalate are commercially available with molecular weights in the range 20 000–200 000. Typical average values are 80 000–130 000.(1)
Hypromellose phthalate is widely used in oral pharmaceutical formulations as an enteric coating material for tablets or granules.(2–8) Hypromellose phthalate is insoluble in gastric fluid but will swell and dissolve rapidly in the upper intestine. Generally, concentrations of 5–10% of hypromellose phthalate are employed with the material being dissolved in either a dichloromethane : ethanol (50 : 50) or an ethanol : water (80 : 20) solvent mixture. Hypromellose phthalate can normally be applied to tablets and granules without the addition of a plasticizer or other film formers, using established coating techniques. However, the addition of a small amount of plasticizer or water can avoid film cracking problems; many commonly used plasticizers, such as diacetin, triacetin, diethyl and dibutyl phthalate, castor oil, acetyl monoglyceride, and polyethylene glycols, are compatible with hypromellose phthalate. Tablets coated with hypromellose phthalate disintegrate more rapidly than tablets coated with cellulose acetate phthalate. Hypromellose phthalate can be applied to tablet surfaces using a dispersion of the micronized hypromellose phthalate powder in an aqueous dispersion of a suitable plasticizer such as triacetin, triethyl citrate, or diethyl tartrate together with a wetting agent.(9) Hypromellose phthalate may be used alone or in combination with other soluble or insoluble binders in the preparation of granules with sustained drug-release properties; the release rate is pH-dependent. Since hypromellose phthalate is tasteless and insoluble in saliva, it can also be used as a coating to mask the unpleasant taste of some tablet formulations. Hypromellose phthalate has also been co-precipitated with a poorly soluble drug to improve dissolution characteristics.(10)
Hypromellose phthalate occurs as white to slightly off-white, freeflowing flakes or as a granular powder. It is odorless or with a slightly acidic odor and has a barely detectable taste.

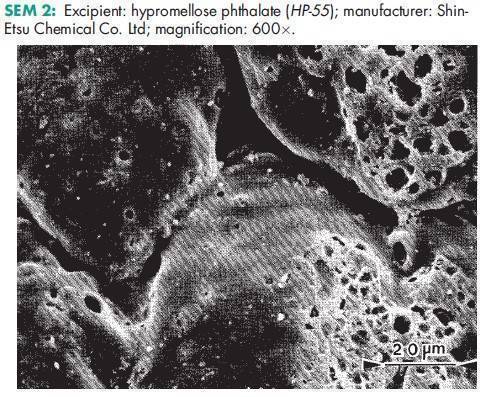
See Table I.
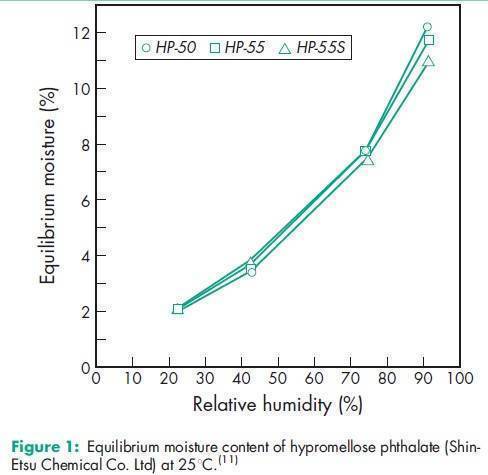
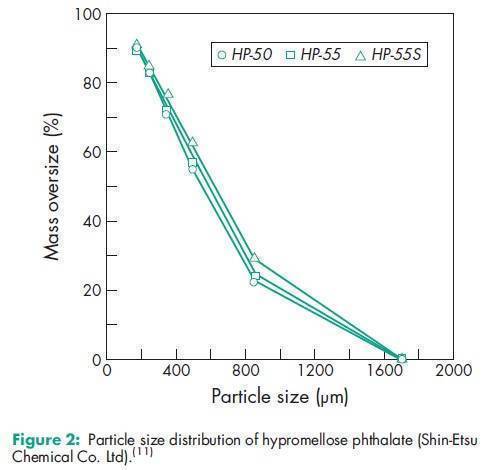
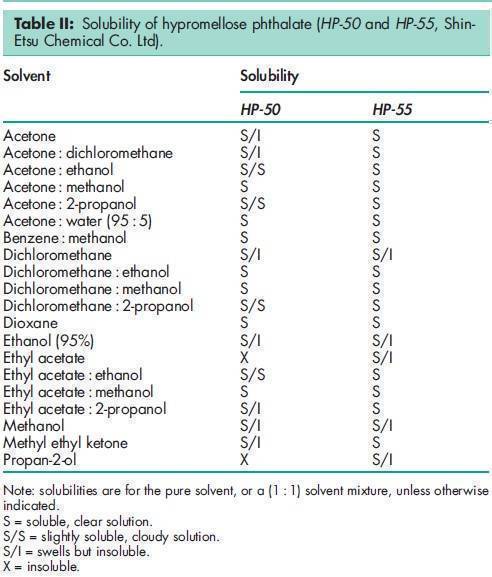
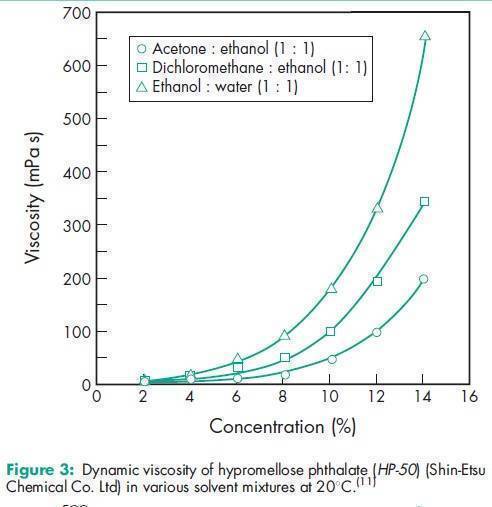
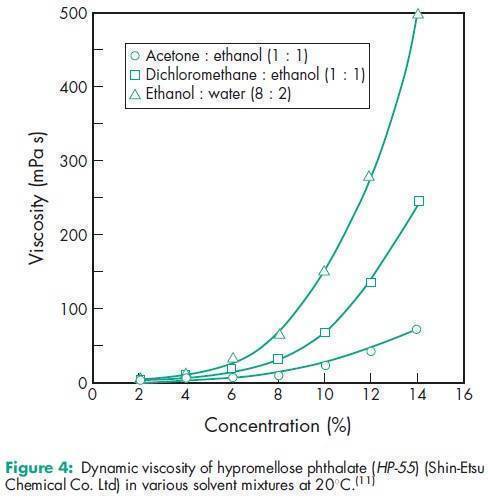
Angle of repose 378 for HP-50; 398 for HP-55; 388 for HP-55S.(11) Density 1.82 g/cm3 for HP-50; 1.65 g/cm3 for HP-55.(11) Density (bulk) 0.278 g/cm3 for HP-50; 0.275 g/cm3 for HP-55; 0.239 g/cm3 for HP-55S.(11) Density (tapped) 0.343 g/cm3 for HP-50; 0.306 g/cm3 for HP-55; 0.288 g/cm3 for HP-55S.(11) Melting point 1508C. Glass transition temperature is 1378C for HP-50 and 1338C for HP-55.(12) Moisture content Hypromellose phthalate is hygroscopic; it takes up 2–5% of moisture at ambient temperature and humidity conditions. For the moisture sorption isotherm of HP-50 measured at 258C, see Figure 1. Particle size distribution see Figure 2. Solubility Readily soluble in a mixture of acetone and methyl or ethyl alcohol (1 : 1), in a mixture of methyl alcohol and dichloromethane (1 : 1), and in aqueous alkali. Practically insoluble in water and dehydrated alcohol and very slightly soluble in acetone. The solubilities of the HP-50 and HP-55 grades, in various solvents and solvent mixtures, are shown in Table II.(11) Viscosity see Figures 3 and 4.
Hypromellose phthalate is chemically and physically stable at ambient temperature for at least 3–4 years and for 2–3 months at 408C and 75% relative humidity.(11) It is stable on exposure to UV light for up to 3 months at 258C and 70% relative humidity. Drums stored in a cool, dry place should be brought to room temperature before opening to prevent condensation of moisture on inside surfaces. After 10 days at 608C and 100% relative humidity, 8–9% of carbyoxybenzoyl group were hydrolyzed. In general, hypromellose phthalate is more stable than cellulose acetate phthalate. At ambient storage conditions, hypromellose phthalate is not susceptible to microbial attack.
Incompatible with strong oxidizing agents. Splitting of film coatings has been reported rarely, most notably with coated tablets that contain microcrystalline cellulose and calcium carboxymethylcellulose. Film splitting has also occurred when a mixture of acetone : propan-2-ol or dichloromethane : pro-pan-2-ol has been used as the coating solvent, or when coatings have been applied in conditions of low temperature and humidity. However, film splitting may be avoided by careful selection of formulation composition, including solvent, by use of a higher molecular weight grade of polymer, or by suitable selection of plasticizer. The addition of more than about 10% titanium dioxide to a coating solution of hypromellose phthalate, which is used to produce a colored film coating, may result in coating with decreased elasticity and resistance to gastric fluid.(11)
Hypromellose phthalate is prepared by the esterification of hypromellose with phthalic anhydride. The degree of alkyloxy and carboxybenzoyl substitution determines the properties of the polymer and in particular the pH at which it dissolves in aqueous media.
Hypromellose phthalate is widely used, primarily as an enteric coating agent, in oral pharmaceutical formulations. Chronic and acute animal feeding studies on several different species have shown no evidence of teratogenicity or toxicity associated with hypromellose phthalate.(13–17) Hypromellose phthalate is generally regarded as a nonirritant and nontoxic material. LD50 (rat, oral): >15 g/kg(13)
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended. Although no threshold limit value has been set for hypromellose phthalate, it should be handled in a well-ventilated environment and the generation of dust should be minimized.
Included in the FDA Inactive Ingredients Database (oral capsules and tablets). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Cellulose acetate phthalate; hypromellose.
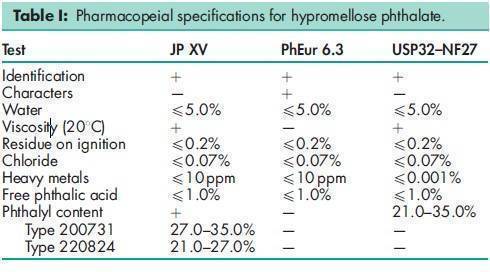
Hypromellose phthalate is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State ofWork’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. Various grades of hypromellose phthalate are available with differing degrees of substitution and physical properties, e.g. grades HP-50, HP-55, and HP-55S (Shin-Etsu Chemical Co. Ltd). See Table III. The number following ‘HP’ in each grade designation refers to the pH value (x10) at which the polymer dissolves in aqueous buffer solutions. The designation ‘S’ in HP-55S indicates a higher molecular weight grade, which produces films with a greater resistance to cracking. In the USA, the substitution type is indicated by a six digit number: the first two digits represent the approximate percentage content of methoxy groups; the next two digits represent the approximate percentage content of hydroxypropoxy groups; and the final two digits represent the approximate percentage content of phthalyl groups. To dissolve hypromellose phthalate in acetone : ethanol (95%) or dichloromethane : alcohol solvent systems, the hypromellose phthalate should first be well dispersed in alcohol before adding acetone or dichloromethane. When using acetone : dichloromethane, hypromellose phthalate should be first dispersed in the dichloromethane and then the acetone added to the system.