

Name: Simethicone
CAS No: a-(Trimethysilyl-o-methylpoly[oxy(dimethylsilylene)], mixture with silicon dioxide [8050-81-5]
BP: Simeticone PhEur: Simeticone USP: Simethicone
Dow Corning Q7-2243 LVA; Dow Corning Q7-2587; polydimethylsiloxane–silicon dioxide mixture; Sentry Simethicone; simeticonum
a-(Trimethysilyl-o-methylpoly[oxy(dimethylsilylene)], mixture with silicon dioxide [8050-81-5]
See Section 8.
where n = 200–350
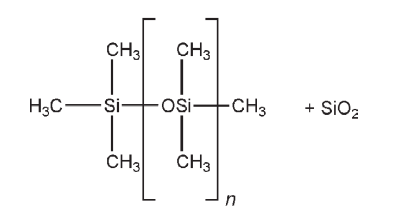
The main use of simethicone as an excipient is as an antifoaming agent in pharmaceutical manufacturing processes, for which 1–50 ppm is used. Therapeutically, simethicone is included in a number of oral pharmaceutical formulations as an antiflatulent, although its therapeutic benefit is questionable.(1,2) It is also included in antacid products such as tablets or capsules.(3–7) In some types of surgical or gastroscopic procedures where gas is used to inflate the body cavity, a defoaming preparation containing simethicone may be used in the area to control foaming of the fluids. When simethicone is used in aqueous formulations, it should be emulsified to ensure compatibility with the aqueous system and components. In the USA, up to 10 ppm of simethicone may be used in food products
The PhEur 6.0 and USP 32 describe simethicone as a mixture of fully methylated linear siloxane polymers containing repeating units of the formula [–(CH3)2SiO–]n, stabilized with trimethylsiloxy endblocking units of the formula [(CH3)3 SiO–], and silicon dioxide. It contains not less than 90.5% and not more than 99.0% of the polydimethylsiloxane [–(CH3)2SiO–]n, and not less than 4.0% and not more than 7.0% of silicon dioxide. The PhEur 6.0 additionally states that the degree of polymerization is between 20–400. Simethicone occurs as a translucent, gray-colored, viscous fluid. It has a molecular weight of 14 000–21 000
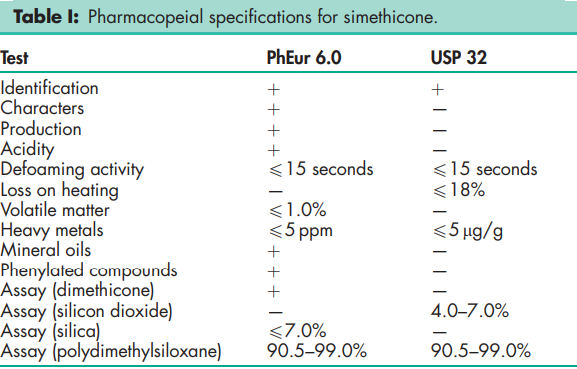
See Table I.
Boiling point 358C Refractive index nD 20 = 0.965–0.970 Solubility Practically insoluble in ethanol (95%) and water. The liquid phase is soluble in benzene, chloroform, and ether, but silicon dioxide remains as a residue in these solvents. Specific gravity 0.95–0.98 at 258C Viscosity (kinematic) 370 mm2 /s (370 cSt) at 258C for Dow Corning Q7-2243 LVA.
Simethicone is generally regarded as a stable material when stored in the original unopened container. A shelf-life of 18 months from the date of manufacture is typical. However, some simethicone products have a tendency for the silicon dioxide to settle slightly and containers of simethicone should therefore be shaken thoroughly to ensure uniformity of contents before sampling or use. Simethicone should be stored in a cool, dry, location away from oxidizing materials. Simethicone can be sterilized by dry heating or autoclaving. With dry heating, a minimum of 4 hours at 1608C is required.
Simethicone as supplied is not generally compatible with aqueous systems and will float like an oil on a formulation unless it is first emulsified. It should not be used in formulations or processing conditions that are very acidic (below pH 3) or highly alkaline (above pH 10), since these conditions may have some tendency to break the polydimethylsiloxane polymer. Simethicone cannot normally be mixed with polar solvents of any kind because it is very minimally soluble. Simethicone is incompatible with oxidizing agents.
Silicon dioxide is initially rendered hydrophobic in one of a variety of proprietary processes specific to a particular manufacturer. It is then slowly mixed with the silicone fluids in a formulation. After mixing, the simethicone is milled to ensure uniformity
Simethicone is used in cosmetics, foods, and oral and topical pharmaceutical formulations, and is generally regarded as a relatively nontoxic and nonirritant material when used as an excipient. Direct contact with the eye may cause irritation. Therapeutically, oral doses of 125–250 mg of simethicone, three or four times daily, have been given as an antiflatulent. Doses of 20–40 mg of simethicone have been given with feeds to relieve colic in infants.(8) LD50 (dog, IV): 0.9 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended. Simethicone should be handled in areas with adequate ventilation.
GRAS listed. Included in the FDA Inactive Ingredients Database (oral emulsions, powders, solutions, suspensions, tablets; and rectal and topical preparations). Included in nonparenteral medicines licensed in the UK.
Cyclomethicone; dimethicone.
The PubChem Compound ID (CID) for simethicone includes 6433516 and 9794495.