

Name: Tagatose
CAS No: (3S,4S,5R)-1,3,4,5,6-pentahydroxyhexan-2-one [87-81-0]
USP-NF: Tagatose
D-lyxo-Hexulose; (3S,4S,5R)-2-(hydroxymethyl)oxane-2,3,4,5- tetrol; Naturlose; D-tagatose; tagatosum; tagatoza.
(3S,4S,5R)-1,3,4,5,6-pentahydroxyhexan-2-one [87-81-0]
C6H12O6 180.16
Tagatose is used as a sweetening agent in beverages, foods, and pharmaceutical applications. A 10% solution of tagatose is about 92% as sweet as a 10% sucrose solution.(1) It is a low-calorie sugar with approximately 38% of the calories of sucrose per gram. It occurs naturally in low levels in milk products.(1) Like other sugars (fructose, glucose, sucrose), it is also used as a bulk sweetener, humectant, texturizer, and stabilizer, and may be used in dietetic foods with a low glycemic index.(2,3)
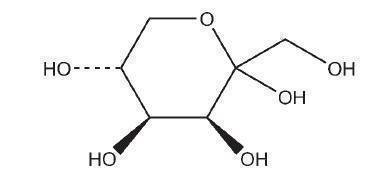
Tagatose is a white, anhydrous crystalline solid. It is a carbohydrate, a ketohexose, an epimer of D-fructose inverted at C-4. It can exist in several tautomeric forms.(4,5)
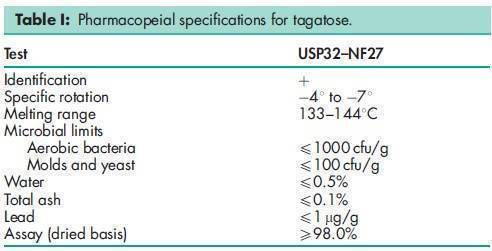
See Table I.
Hygroscopicity Crystalline D-tagatose has low hygroscopicity similar to that of sucrose. Melting point 132–1358C Solubility Very soluble in water: 1 in 0.7 parts water (58% w/w) at 218C. Slightly soluble in ethanol: 1 in 5000 parts ethanol.
Tagatose is stable under pH conditions typically encountered in foods (pH>3). It is a reducing sugar and undergoes the Maillard reaction. Tagatose is stable under typical storage conditions. It caramelizes at elevated temperature.
A Maillard-type condensation reaction is likely to occur between tagatose and compounds with a primary amine group to form brown or yellow-brown colored Amidori compounds. Reducing sugars will also interact with secondary amines to form an imine, but without any accompanying yellow-brown discoloration.
Tagatose is obtained from D-galactose by isomerization under alkaline conditions in the presence of calcium.
Tagatose is safe for use in food and beverages. It has been used in pharmaceutical products.(1)
Observe normal precautions appropriate to the circumstances and quantity of the material handled. Excessive generation of dust, or inhalation of dust, should be avoided.
GRAS listed. Included in the FDA Inactive Ingredients Database (oral and rectal solutions).
DL-Tagatose; L-tagatose. DL-Tagatose Empirical formula C6H12O6 CAS number [17598-81-1] Synonyms lyxo-2-Hexulose L-Tagatose Empirical formula C6H12O6 CAS number [17598-82-2] Melting point 134–1358C Specific rotation aD 16 = þ18 (2% aqueous solution) Comments Sweetening agent for pharmaceutical and personal aid products.
The EINECS number for tagatose is 201-772-3. The PubChem Compound ID (CID) for tagatose is 92092.