

Name: Talc
CAS No: Talc [14807-96-6]
BP: Purified Talc JP: Talc PhEur: Talc USP: Talc
Altalc; E553b; hydrous magnesium calcium silicate; hydrous magnesium silicate; Imperial; Luzenac Pharma; magnesium hydrogen metasilicate; Magsil Osmanthus; Magsil Star; powdered talc; purified French chalk; Purtalc; soapstone; steatite; Superiore; talcum.
Talc [14807-96-6]
Talc is a purified, hydrated, magnesium silicate, approximating to the formula Mg6(Si2O5)4(OH)4. It may contain small, variable amounts of aluminum silicate and iron.
Talc was once widely used in oral solid dosage formulations as a lubricant and diluent, see Table I,(1–3) although today it is less commonly used. However, it is widely used as a dissolution retardant in the development of controlled-release products.(4–6) Talc is also used as a lubricant in tablet formulations;(7) in a novel powder coating for extended-release pellets;(8) and as an adsorbant.( 9) In topical preparations, talc is used as a dusting powder, although it should not be used to dust surgical gloves; see Section 14. Talc is a natural material; it may therefore frequently contain microorganisms and should be sterilized when used as a dusting powder; see Section 11. Talc is additionally used to clarify liquids and is also used in cosmetics and food products, mainly for its lubricant properties.
Talc is a very fine, white to grayish-white, odorless, impalpable, unctuous, crystalline powder. It adheres readily to the skin and is soft to the touch and free from grittiness.
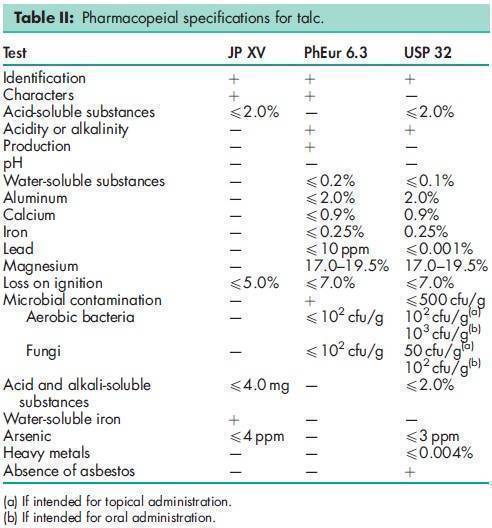
See Table II.
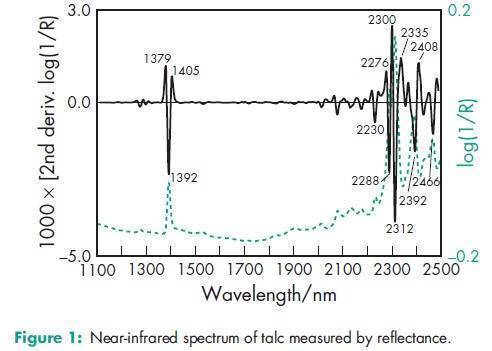
Acidity/alkalinity pH = 7–10 for a 20% w/v aqueous dispersion. Hardness (Mohs) 1.0–1.5 Moisture content Talc absorbs insignificant amounts of water at 258C and relative humidities up to about 90%. NIR spectra see Figure 1. Particle size distribution Varies with the source and grade of material. Two typical grades are 599% through a 74 mm (#200 mesh) or 599% through a 44 mm (#325 mesh). Refractive index nD 20 = 1.54–1.59 Solubility Practically insoluble in dilute acids and alkalis, organic solvents, and water. Specific gravity 2.7–2.8 Specific surface area 2.41–2.42m2/g



Talc is a stable material and may be sterilized by heating at 1608C for not less than 1 hour. It may also be sterilized by exposure to ethylene oxide or gamma irradiation.(10) Talc should be stored in a well-closed container in a cool, dry place.
Incompatible with quaternary ammonium compounds.
Talc is a naturally occurring hydropolysilicate mineral found in many parts of the world including Australia, China, Italy, India, France, and the USA.(11) The purity of talc varies depending on the country of origin. For example, Italian types are reported to contain calcium silicate as the contaminant; Indian types contain aluminum and iron oxides; French types contain aluminum oxide; and American types contain calcium carbonate (California), iron oxide (Montana), aluminum and iron oxides (North Carolina), or aluminum oxide (Alabama).( 12) Naturally occurring talc is mined and pulverized before being subjected to flotation processes to remove various impurities such as asbestos (tremolite); carbon; dolomite; iron oxide; and various other magnesium and carbonate minerals. Following this process, the talc is finely powdered, treated with dilute hydrochloric acid, washed with water, and then dried. The processing variables of agglomerated talc strongly influence its physical characteristics.( 13–15)
Talc is used mainly in tablet and capsule formulations. Talc is not absorbed systemically following oral ingestion and is therefore regarded as an essentially nontoxic material. However, intranasal or intravenous abuse of products containing talc can cause granulomas in body tissues, particularly the lungs.(16–18) Contamination of wounds or body cavities with talc may also cause granulomas; therefore, it should not be used to dust surgical gloves. Inhalation of talc causes irritation and may cause severe respiratory distress in infants;(19) see also Section 15. Although talc has been extensively investigated for its carcinogenic potential, and it has been suggested that there is an increased risk of ovarian cancer in women using talc, the evidence is inconclusive.(20,21) However, talc contaminated with asbestos has been proved to be carcinogenic in humans, and asbestos-free grades should therefore be used in pharmaceutical products.(22) Also, long-term toxic effects of talc contaminated with large quantities of hexachlorophene caused serious irreversible neurotoxicity in infants accidentally exposed to the substance.(23)
Observe normal precautions appropriate to the circumstances and quantity of material handled. Talc is irritant if inhaled and prolonged excessive exposure may cause pneumoconiosis. In the UK, the workplace exposure limit for talc is 1 mg/m3 of respirable dust long-term (8-hour TWA).(24) Eye protection, gloves, and a respirator are recommended.
Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (buccal tablets; oral capsules and tablets; rectal and topical preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Bentonite; magnesium aluminum silicate; magnesium silicate; magnesium trisilicate.
Talc is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32– NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. Various grades of talc are commercially available that vary in their chemical composition depending upon their source and method of preparation.(11,25,26) Talc derived from deposits that are known to contain associated asbestos is not suitable for pharmaceutical use. Tests for amphiboles and serpentines should be carried out to ensure that the product is free of asbestos. A specification for talc is contained in the Food Chemicals Codex (FCC).(27) The EINECS number for talc is 238-877-9. The PubChem Compound ID (CID) for talc includes 26924, 443754 and 16211421.