

Name: Tartaric Acid
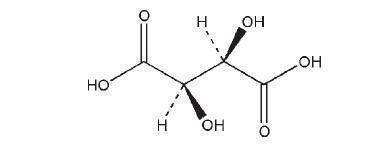
CAS No: [R-(R*,R*)]-2,3-Dihydroxybutanedioic acid [87-69-4]
BP: Tartaric Acid JP: Tartaric Acid PhEur: Tartaric Acid USP-NF: Tartaric Acid
Acidum tartaricum; L-(þ)-2,3-dihydroxybutanedioic acid; (2R,3R)- 2,3-dihydroxybutane-1,4-dioic acid; 2,3-dihydroxysuccinic acid; E334; d-tartaric acid; L-(þ)-tartaric acid.
[R-(R*,R*)]-2,3-Dihydroxybutanedioic acid [87-69-4]
C4H6O6 150.09
Tartaric acid is used in beverages, confectionery, food products, and pharmaceutical formulations as an acidulant. It may also be used as a sequestering agent and as an antioxidant synergist. In pharmaceutical formulations, it is widely used in combination with bicarbonates, as the acid component of effervescent granules, powders, and tablets. Tartaric acid is also used to form molecular compounds (salts and cocrystals) with active pharmaceutical ingredients to improve physicochemical properties such as dissolution rate and solubility.( 1,2)
Tartaric acid occurs as colorless monoclinic crystals, or a white or almost white crystalline powder. It is odorless, with an extremely tart taste.
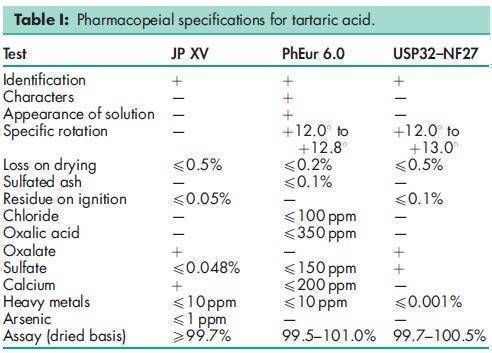
See Table I.
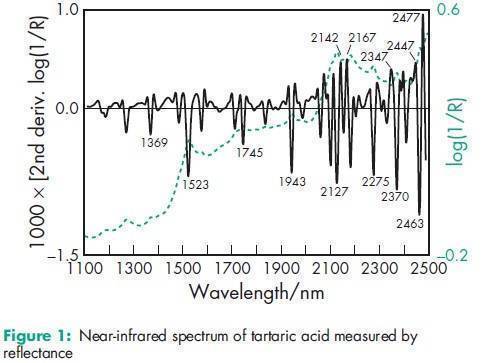
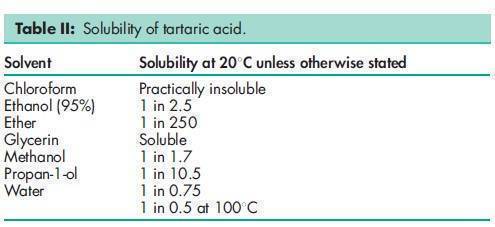
Acidity/alkalinity pH = 2.2 (1.5% w/v aqueous solution) Density 1.76 g/cm3 Dissociation constant pKa1 = 2.93 at 258C; pKa2 = 4.23 at 258C. Heat of combustion 1151 kJ/mol (275.1 kcal/mol) Melting point 168–1708C NIR spectra see Figure 1. Osmolarity A 3.9% w/v aqueous solution is isoosmotic with serum. Solubility see Table II. Specific heat 1.20 J/g (0.288 cal/g) at 208C. Specific rotation [a]D 20 =þ12.08 (20% w/v aqueous solution).
The bulk material is stable and should be stored in a well-closed container in a cool, dry place.
Tartaric acid is incompatible with silver and reacts with metal carbonates and bicarbonates (a property exploited in effervescent preparations).
Tartaric acid occurs naturally in many fruits as the free acid or in combination with calcium, magnesium, and potassium. Commercially, L-(þ)-tartaric acid is manufactured from potassium tartrate (cream of tartar), a by-product of wine making. Potassium tartrate is treated with hydrochloric acid, followed by the addition of a calcium salt to produce insoluble calcium tartrate. This precipitate is then removed by filtration and reacted with 70% sulfuric acid to yield tartaric acid and calcium sulfate.
Tartaric acid is widely used in food products and oral, topical, and parenteral pharmaceutical formulations. It is generally regarded as a nontoxic and nonirritant material; however, strong tartaric acid solutions are mildly irritant and if ingested undiluted may cause gastroenteritis. An acceptable daily intake for L-(þ)-tartaric acid has not been set by the WHO, although an acceptable daily intake of up to 30 mg/kg body-weight for monosodium L-(þ)-tartrate has been established.(3) LD50 (mouse, IV): 0.49 g/kg(4)
Observe normal precautions appropriate to the circumstances and quantity of material handled. Tartaric acid may be irritant to the eyes; eye protection and rubber or plastic gloves are recommended. When heated to decomposition, tartaric acid emits acrid smoke and fumes.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (IM and IV injections; oral solutions, syrups and tablets; sublingual tablets; topical films; rectal and vaginal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Citric acid monohydrate; fumaric acid; malic acid.
L-(þ)-tartaric acid, the optical isomer usually encountered, is the naturally occurring form and is specified as tartaric acid in the PhEur 6.0 and USP32–NF27. A specification for tartaric acid is contained in the Food Chemicals Codex (FCC).(5) The EINECS number for tartaric acid is 201-766-0.