

Name: Sodium Ascorbate
CAS No: Monosodium L-(þ)-ascorbate [134-03-2]
BP: Sodium Ascorbate PhEur: Sodium Ascorbate USP: Sodium Ascorbate
L-Ascorbic acid monosodium salt; E301; 3-oxo-L-gulofuranolactone sodium enolate; natrii ascorbas; SA-99; vitamin C sodium.
Monosodium L-(þ)-ascorbate [134-03-2]
C6H7NaO6 198.11
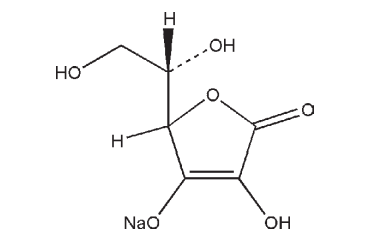
Sodium ascorbate is used as an antioxidant in pharmaceutical formulations, and also in food products where it increases the effectiveness of sodium nitrite against growth of Listeria monocytogenes in cooked meats. It improves gel cohesiveness and sensory firmness of fiberized products regardless of vacuum treatment. It is also used therapeutically as a source of vitamin C in tablets and parenteral preparations.
Sodium ascorbate occurs as a white or slightly yellow-colored, practically odorless, crystalline powder with a pleasant saline taste.
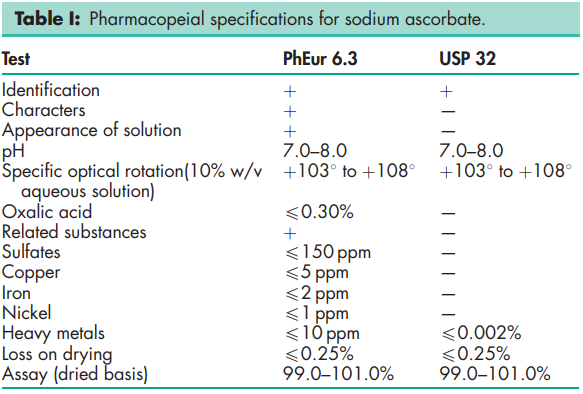
See Table I.
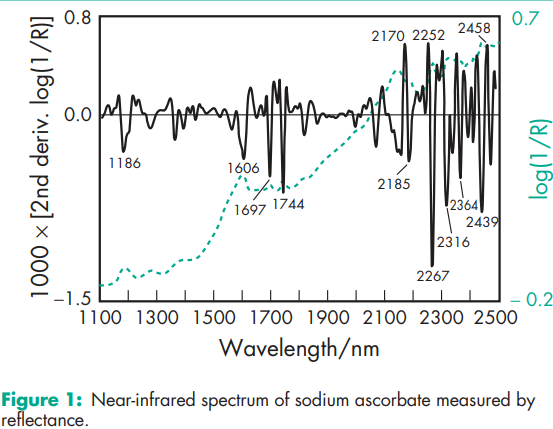
Acidity/alkalinity pH = 7–8 (10% w/v aqueous solution) Density (tapped) 0.6–1.1 g/cm3 for fine powder; 0.8–1.1 g/cm3 for fine granular grade. Density (true) 1.826 g/cm3 Hygroscopicity Not hygroscopic. Sodium ascorbate adsorbs practically no water up to 80% relative humidity at 208C and less than 1% w/w of water at 90% relative humidity. Melting point 2188C (with decomposition) NIR spectra see Figure 1. Particle size distribution Various grades of sodium ascorbate with different particle-size distributions are commercially available, e.g. approximately 98% passes through a 149 mm mesh for a fine powder grade (Takeda), and approximately 95% passes through a 840 mm mesh for a standard grade (Takeda). Solubility see Table II. Specific gravity 1.782 for powder at 208C; 1.005 for 1% w/v aqueous solution at 258C; 1.026 for 5% w/v aqueous solution at 258C. Specific rotation [a]D 20 = þ104.48 (10% w/v aqueous solution)


Sodium ascorbate is relatively stable in air, although it gradually darkens on exposure to light. Aqueous solutions are unstable and subject to rapid oxidation in air at pH > 6.0. The bulk material should be stored in a well-closed nonmetallic container, protected from light, in a cool, dry place.
Incompatible with oxidizing agents, heavy metal ions, especially copper and iron, methenamine, sodium nitrite, sodium salicylate, and theobromine salicylate. The aqueous solution is reported to be incompatible with stainless steel filters.(1)
An equivalent amount of sodium bicarbonate is added to a solution of ascorbic acid in water. Following the cessation of effervescence, the addition of propan-2-ol precipitates sodium ascorbate
The parenteral administration of 0.251.00 g of sodium ascorbate, given daily in divided doses, is recommended in the treatment of vitamin C deficiencies. Various adverse reactions have been reported following the administration of 1 g or more of sodium ascorbate, although ascorbic acid and sodium ascorbate are usually well tolerated; see Ascorbic acid. There have been no reports of adverse effects associated with the much lower concentrations of sodium ascorbate and ascorbic acid, which are employed as antioxidants. The WHO has set an acceptable daily intake of ascorbic acid, potassium ascorbate, and sodium ascorbate, as antioxidants in food, at up to 15 mg/kg body-weight in addition to that naturally present in food.(2)
Observe normal precautions appropriate to the circumstances and quantity of material handled. Sodium ascorbate may be irritant to the eyes. Eye protection and rubber or plastic gloves are recommended.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (IV preparations; oral tablets). Included in nonparenteral and parenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Ascorbic acid; ascorbyl palmitate; calcium ascorbate. Calcium ascorbate Empirical formula C12H14O12Ca Molecular weight 390.31 CAS number [5743-27-1] Synonyms calcium L-(þ)-ascorbate; CCal-97; E302.
1 mg of sodium ascorbate is equivalent to 0.8890 mg of ascorbic acid (1 mg of ascorbic acid is equivalent to 1.1248 mg of sodium ascorbate); 1 g of sodium ascorbate contains approximately 5 mmol of sodium. A specification for sodium ascorbate is contained in the Food Chemicals Codex (FCC).(3) The EINECS number for sodium ascorbate is 205-126-1. The PubChem Compound ID (CID) for sodium ascorbate is 23666832.