

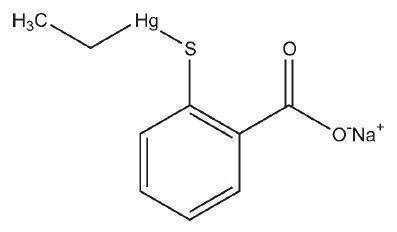
Name: Thimerosal
CAS No: Ethyl[2-mercaptobenzoato(2–)-O,S]-mercurate(1–) sodium [54- 64-8]
BP: Thiomersal PhEur: Thiomersal USP: Thimerosal
[(o-Carboxyphenyl)thio]ethylmercury sodium salt; ethyl (2-mercaptobenzoato- S)-mercury, sodium salt; ethyl (sodium o-mercaptobenzoato) mercury; mercurothiolate; sodium ethylmercurithiosalicylate; Thimerosal Sigmaultra; thiomersalate; thiomersalum.
Ethyl[2-mercaptobenzoato(2–)-O,S]-mercurate(1–) sodium [54- 64-8]
C9H9HgNaO2S 404.81
Thimerosal has been used as an antimicrobial preservative in biological and pharmaceutical preparations since the 1930s;(1) see Table I. It is used as an alternative to benzalkonium chloride and other phenylmercuric preservatives, and has both bacteriostatic and fungistatic activity. Increasing concerns over its safety have, however, led to questions regarding its continued use in formulations; Thimerosal is also used in cosmetics (see Section 16) and to preserve soft contact lens solutions. Therapeutically, thimerosal is occasionally used as a bacteriostatic and fungistatic mercurial antiseptic, which is usually applied topically at a concentration of 0.1% w/w.(2) However, its use is declining owing to its toxicity and effects on the environment.
Thimerosal is a light cream-colored crystalline powder with a slight, characteristic odor.
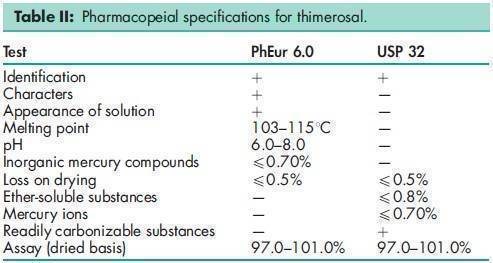
See Table II.
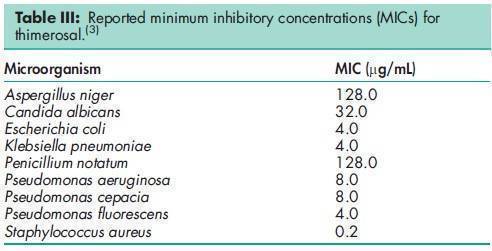
Acidity/alkalinity pH = 6.7 for a 1% w/v aqueous solution at 208C. Antimicrobial activity Thimerosal is bactericidal at acidic pH, bacteriostatic and fungistatic at alkaline or neutral pH. Thimerosal is not effective against spore-forming organisms. See also Section 12. For reported minimum inhibitory concentrations (MICs), see Table III.(3) Density (bulk) <0.33 g/cm3 Dissociation constant pKa = 3.05 at 258C. Melting point 232–2338C with decomposition. Solubility Soluble 1 in 8 of ethanol (95%), 1 in 1 of water; practically insoluble in benzene and ether.
Thimerosal is stable at normal temperatures and pressures; exposure to light may cause discoloration. Aqueous solutions may be sterilized by autoclaving but are sensitive to light. The rate of oxidation in solutions is increased by the presence of trace amounts of copper and other metals. Edetic acid or edetates may be used to stabilize solutions but have been reported to reduce the antimicrobial efficacy of thimerosal solutions; see Section 12. The solid material should be stored in a well-closed container, protected from light, in a cool, dry place.
Incompatible with aluminum and other metals, strong oxidizing agents, strong acids and bases, sodium chloride solutions,(4) lecithin, phenylmercuric compounds, quaternary ammonium compounds, thioglycolate, and proteins. The presence of sodium metabisulfite, edetic acid, and edetates in solutions can reduce the preservative efficacy of thimerosal.(5) In solution, thimerosal may be adsorbed by plastic packaging materials, particularly polyethylene. It is strongly adsorbed by treated or untreated rubber caps that are in contact with solutions.(6,7) When it was used with cyclodextrin, the effectiveness of thimerosal was reduced; however, this was related to the lipid nature of the other ingredients in the preparation.(8)
Thimerosal is prepared by the interaction of ethylmercuric chloride, or hydroxide, with thiosalicylic acid and sodium hydroxide, in ethanol (95%).
Thimerosal is widely used as an antimicrobial preservative in parenteral and topical pharmaceutical formulations. However, concern over the use of thimerosal in pharmaceuticals has increased as a result of a greater awareness of the toxicity of mercury and other associated mercury compounds.(9,10) The increasing number of reports of adverse reactions, particularly hypersensitivity,(11–13) to thimerosal and doubts as to its effectiveness as a preservative have led to suggestions that it should not be used as a preservative in eye drops(14) or vaccines.(15–17) In both Europe and the USA, regulatory bodies have recommended that thimerosal in vaccines be phased out.(18–20) More recent studies assessing the safety of thimerosal in vaccines have, however, suggested that while the risk of hypersensitivity reactions is present, the relative risk of neurological harm in infants is negligible given the quantities of thimerosal present in vaccines.(21–23) Regulatory bodies in Europe and the USA have therefore updated their advice on the use of thimerosal in vaccines by stating that while it would be desirable for thimerosal not to be included in vaccines and other formulations the benefits of vaccines far outweigh any risks of adverse effects associated with their use.(24–27) The most frequently reported adverse reaction to thimerosal, particularly in vaccines,(15–30) is hypersensitivity, usually with erythema and papular or vesicular eruptions. Although not all thimerosal-sensitive patients develop adverse reactions to vaccines containing thimerosal, there is potential risk. Patch testing in humans and animal experiments have suggested that 0.1% w/v thimerosal can sensitize children.(31) The incidence of sensitivity to thimerosal appears to be increasing; a study of 256 healthy subjects showed approximately 6% with positive sensitivity.(32) Adverse reactions to thimerosal used to preserve contact lens solutions have also been reported. Reactions include ocular redness, irritation, reduced lens tolerance, and conjunctivitis.(33–35) One estimate suggests that approximately 10% of contact lens wearers may be sensitive to thimerosal.(36) Thimerosal has also been associated with false positive reactions to old tuberculin,(37) ototoxicity,(38) and an unusual reaction to aluminum(39) in which a patient suffered a burn 5 cm in diameter at the site of an aluminum foil diathermy electrode after preoperative preparation of the skin with a 0.1% w/v thimerosal solution in ethanol (50%). Investigation showed that considerable heat was generated when such a solution came into contact with aluminum. An interaction between orally administered tetracyclines and thimerosal, which resulted in varying extents of ocular irritation, has been reported in patients using a contact lens solution preserved with thimerosal.(40) Controversially, some have claimed a connection between the use of thimerosal in vaccines and the apparent rise in the incidence of autism. However, recent studies have shown no association between thimerosal exposure and autism.(24–27,41,42) Serious adverse effects and some fatalities have been reported following the parenteral and topical use of products containing thimerosal. Five fatal poisonings resulted from the use of 1000 times the normal concentration of thimerosal in a chloramphenicol preparation for intramuscular injection.(43) Ten out of 13 children died as a result of treatment of umbilical hernia (omphaloceles) with a topical tincture of thimerosal.(44) It has therefore been recommended that organic mercurial disinfectants should be restricted or withdrawn from use in hospital since absorption occurs readily through intact membranes. In a case of attempted suicide, a 44-year-old man drank 83 mg/kg of a thimerosal-containing solution. Despite spontaneously vomiting after 15 minutes, gastric lavage and administration of chelating agents on hospital admission, serious symptoms ultimately ending in coma occurred. The patient survived and after 5 months treatment made a full recovery except for sensory defects in two toes.(45) LD50 (mouse, oral): 91 mg/kg(46) LD50 (rat, oral): 75 mg/kg LD50 (rat, SC): 98 mg/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Thimerosal is irritant to the skin and mucous membranes, and may be systemically absorbed through the skin and upper respiratory tract. Thimerosal should be handled in a well-ventilated environment. Eye protection, gloves, and a respirator are recommended. Chemical decomposition may cause the release of toxic fumes containing oxides of carbon, sulfur, and mercury in addition to mercury vapor.
Included in the FDA Inactive Ingredients Database (IM, IV, and SC injections; ophthalmic, otic, and topical preparations). Included in nonparenteral and parenteral medicines licensed in the UK. In the UK, the use of thimerosal in cosmetics is limited to 0.003% w/w (calculated as mercury) as a preservative in shampoos and haircreams, which contain nonionic emulsifiers that would render other preservatives ineffective. The total permitted concentration (calculated as mercury) when mixed with other mercury compounds is 0.007% w/w.(47) Included in the Canadian List of Acceptable Nonmedicinal Ingredients.
Phenylmercuric acetate; phenylmercuric borate; phenylmercuric nitrate.
Some variation between the results obtained when comparing different thimerosal assay methods has been reported.(48,49) The EINECS number for thimerosal is 200-210-4. The PubChem Compound ID (CID) for thiomersal is 16684434.