

Name: Sodium Benzoate
CAS No: Sodium benzoate [532-32-1]
BP: Sodium Benzoate JP: Sodium Benzoate PhEur: Sodium Benzoate USP-NF: Sodium Benzoate
Benzoic acid sodium salt; benzoate of soda; E211; natrii benzoas; natrium benzoicum; sobenate; sodii benzoas; sodium benzoic acid.
Sodium benzoate [532-32-1]
C7H5NaO2 144.11
Sodium benzoate is used primarily as an antimicrobial preservative in cosmetics, foods, and pharmaceuticals. It is used in concentrations of 0.02–0.5% in oral medicines, 0.5% in parenteral products, and 0.1–0.5% in cosmetics. The usefulness of sodium benzoate as a preservative is limited by its effectiveness over a narrow pH range; see Section 10. Sodium benzoate is used in preference to benzoic acid in some circumstances, owing to its greater solubility. However, in some applications it may impart an unpleasant flavor to a product. Sodium benzoate has also been used as a tablet lubricant(1) at 2–5% w/w concentrations. Solutions of sodium benzoate have also been administered, orally or intravenously, in order to determine liver function.
Sodium benzoate occurs as a white granular or crystalline, slightly hygroscopic powder. It is odorless, or with faint odor of benzoin and has an unpleasant sweet and saline taste.
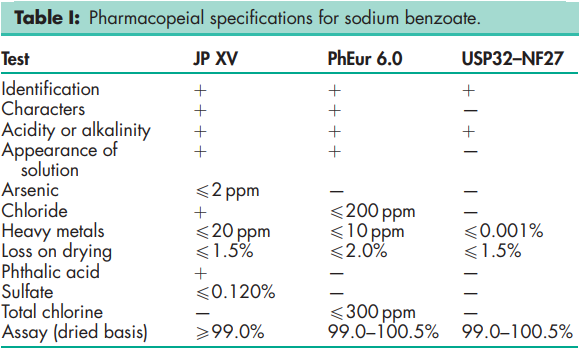
See Table I.
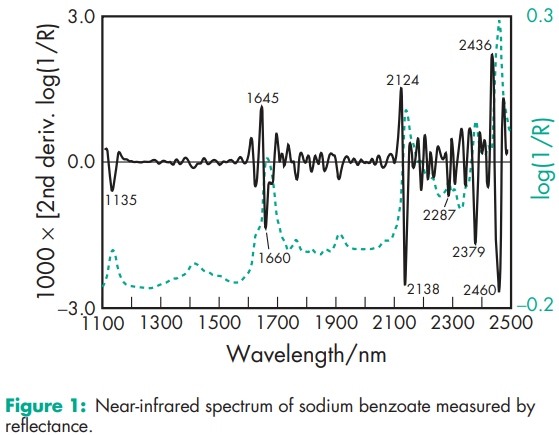
Acidity/alkalinity pH = 8.0 (saturated aqueous solution at 258C). It is relatively inactive above approximately pH 5. Antimicrobial activity Sodium benzoate has both bacteriostatic and antifungal properties attributed to undissociated benzoic acid; hence preservative efficacy is best seen in acidic solutions (pH 2–5). In alkaline conditions it is almost without effect. Density 1.497–1.527 g/cm3 at 248C Freezing point depression 0.248C (1.0% w/v) NIR spectra see Figure 1. Osmolarity A 2.25% w/v aqueous solution is iso-osmotic with serum. Partition coefficients Vegetable oil : water = 3–6 Solubility
Aqueous solutions may be sterilized by autoclaving or filtration.The bulk material should be stored in a well-closed container, in a cool, dry place


Incompatible with quaternary compounds, gelatin, ferric salts, calcium salts, and salts of heavy metals, including silver, lead, and mercury. Preservative activity may be reduced by interactions with kaolin(2) or nonionic surfactants.
Prepared by the treatment of benzoic acid with either sodium carbonate or sodium bicarbonate.
Ingested sodium benzoate is conjugated with glycine in the liver to yield hippuric acid, which is excreted in the urine. Symptoms of systemic benzoate toxicity resemble those of salicylates.(3) Whereas oral administration of the free-acid form may cause severe gastric irritation, benzoate salts are well tolerated in large quantities: e.g. 6 g of sodium benzoate in 200 mL of water is administered orally as a liver function test. Clinical data have indicated that sodium benzoate can produce nonimmunological contact urtcaria and nonimmunological immediate contact reactions.(4) However, it is also recognized that these reactions are strictly cutaneous, and sodium benzoate can therefore be used safely at concentrations up to 5%. However, this nonimmunological phenomenon should be considered when designing formulations for infants and children. Other adverse effects include anaphylaxis(5–7) and urticarial reactions, although a controlled study has shown that the incidence of urticaria in patients given benzoic acid is no greater than that with a lactose placebo.(8) It has been recommended that caffeine and sodium benzoate injection should not be used in neonates;(9) however, sodium benzoate has been used by others in the treatment of some neonatal metabolic disorders.(10) It has been suggested that there is a general adverse effect of benzoate preservatives on the behavior of 3-yearold children, which is detectable by parents, but not by a simple clinical assessment.(11) The WHO acceptable daily intake of total benzoates, calculated as benzoic acid, has been estimated at up to 5 mg/kg of bodyweight.(12,13) LD50 (mouse, IM): 2.3 g/kg(13,14) LD50 (mouse, IV): 1.4 g/kg LD50 (mouse, oral): 1.6 g/kg LD50 (rabbit, oral): 2.0 g/kg LD50 (rat, IV): 1.7 mg/kg LD50 (rat, oral): 4.1 g/kg

Observe normal precautions appropriate to the circumstances and quantity of material handled. Sodium benzoate may be irritant to the eyes and skin. Eye protection and rubber or plastic gloves are recommended.
GRAS listed. Accepted as a food additive in Europe. Included in the FDA Inactive Ingredients Database (dental preparations; IM and IV injections; oral capsules, solutions and tablets; rectal; and topical preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Benzoic acid; potassium benzoate.
Sodium benzoate has been used as an antimicrobial agent used in polymeric films in food packaging.(15) A specification for sodium benzoate is contained in the Food Chemicals Codex (FCC).(16) The EINECS number for sodium benzoate is 208-534-8. The PubChem Compound ID (CID) for sodium benzoate is 517055