

Name: Sodium Bicarbonate
CAS No: Carbonic acid monosodium salt [144-55-8]
BP: Sodium Bicarbonate JP: Sodium Bicarbonate PhEur: Sodium Hydrogen Carbonate USP: Sodium Bicarbonate
Baking soda; E500; Effer-Soda; monosodium carbonate; natrii hydrogenocarbonas; Sal de Vichy; sodium acid carbonate; sodium hydrogen carbonate.
Carbonic acid monosodium salt [144-55-8]
NaHCO3 84.01
See Section 4
Sodium bicarbonate is generally used in pharmaceutical formulations as a source of carbon dioxide in effervescent tablets and granules. It is also widely used to produce or maintain an alkaline pH in a preparation. In effervescent tablets and granules, sodium bicarbonate is usually formulated with citric and/or tartaric acid;(1) combinations of citric and tartaric acid are often preferred in formulations as citric acid alone produces a sticky mixture that is difficult to granulate, while if tartaric acid is used alone, granules lose firmness. When the tablets or granules come into contact with water, a chemical reaction occurs, carbon dioxide is evolved, and the product disintegrates.(2,3) Melt granulation in a fluidized bed dryer has been suggested as a one-step method for the manufacture of effervescent granules composed of anhydrous citric acid and sodium bicarbonate, for subsequent compression into tablets.(4) Tablets may also be prepared with sodium bicarbonate alone since the acid of gastric fluid is sufficient to cause effervescence and disintegration. Sodium bicarbonate is also used in tablet formulations to buffer drug molecules that are weak acids, thereby increasing the rate of tablet dissolution and reducing gastric irritation.(5–7) The effects of tablet binders, such as polyethylene glycols, microcrystalline cellulose, silicified microcrystalline cellulose, pregelatinized starch, and povidone, on the physical and mechanical properties of sodium bicarbonate tablets have also been investigated.(8,9) Additionally, sodium bicarbonate is used in solutions as a buffering agent for erythromycin,(10) lidocaine,(11) local anesthetic solutions,(12) and total parenteral nutrition (TPN) solutions.(13) In some parenteral formulations, e.g. niacin, sodium bicarbonate is used to produce a sodium salt of the active ingredient that has enhanced solubility. Sodium bicarbonate has also been used as a freeze-drying stabilizer(14) and in toothpastes. Recently, sodium bicarbonate has been used as a gas-forming agent in alginate raft systems(15–17) and in floating, controlledrelease oral dosage forms for a range of drugs.(18–27) Tablet formulations containing sodium bicarbonate have been shown to increase the absorption of paracetamol,(28,29) and improve the stability of levothyroxine.(30) Sodium bicarbonate has also been included in formulations of vaginal bioadhesive tablets(31) and in carbon dioxide releasing suppositories.(32) Therapeutically, sodium bicarbonate may be used as an antacid, and as a source of the bicarbonate anion in the treatment of metabolic acidosis. Sodium bicarbonate may also be used as a component of oral rehydration salts and as a source of bicarbonate in dialysis fluids; it has also been suggested as a means of preventing radiocontrast-induced nephrotoxicity.(33) Sodium bicarbonate is used in food products as an alkali or as a leavening agent, e.g. baking soda

Sodium bicarbonate occurs as an odorless, white, crystalline powder with a saline, slightly alkaline taste. The crystal structure is monoclinic prisms. Grades with different particle sizes, from a fine powder to free-flowing uniform granules, are commercially available
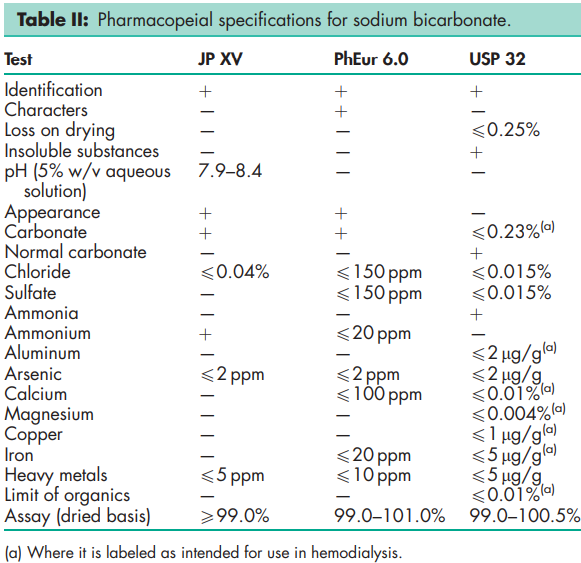
See Table II.
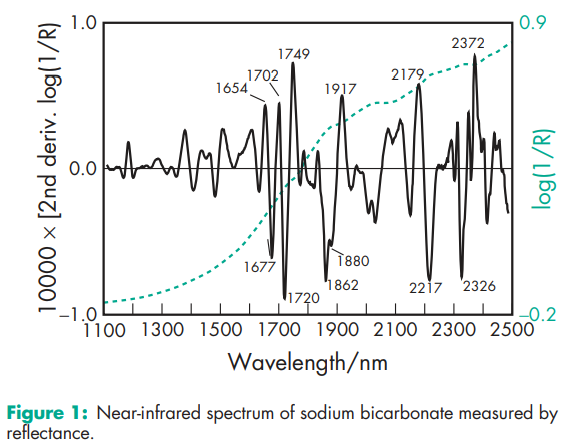
Acidity/alkalinity pH = 8.3 for a freshly prepared 0.1 M aqueous solution at 258C; alkalinity increases on standing, agitation, or heating. Density (bulk) 0.869 g/cm3 Density (tapped) 1.369 g/cm3 Density(true) 2.173 g/cm3 Freezing point depression 0.3818C (1% w/v solution) Melting point 2708C (with decomposition) Moisture content Below 80% relative humidity, the moisture content is less than 1% w/w. Above 85% relative humidity, sodium bicarbonate rapidly absorbs excessive amounts of water and may start to decompose with loss of carbon dioxide. NIR spectra see Figure 1. Osmolarity A 1.39% w/v aqueous solution is isoosmotic with serum. Refractive index nD 20 = 1.3344 (1% w/v aqueous solution) Solubility


When heated to about 508C, sodium bicarbonate begins to dissociate into carbon dioxide, sodium carbonate, and water; on heating to 250–3008C, for a short time, sodium bicarbonate is completely converted into anhydrous sodium carbonate. However, the process is both time- and temperature-dependent, with conversion 90% complete within 75 minutes at 938C. The reaction proceeds via surface-controlled kinetics; when sodium bicarbonate crystals are heated for a short period of time, very fine needleshaped crystals of anhydrous sodium carbonate are formed on the sodium bicarbonate surface.(34) The effects of relative humidity and temperature on the moisture sorption and stability of sodium bicarbonate powder have been investigated. Sodium bicarbonate powder is stable below 76% relative humidity at 258C and below 48% relative humidity at 408C.(35) At 54% relative humidity, the degree of pyrolytic decarboxylation of sodium bicarbonate should not exceed 4.5% in order to avoid detrimental effects on stability.(36) At ambient temperatures, aqueous solutions slowly decompose with partial conversion into the carbonate; the decomposition is accelerated by agitation or heat. Aqueous solutions begin to break up into carbon dioxide and sodium carbonate at about 208C, and completely on boiling. Aqueous solutions of sodium bicarbonate may be sterilized by filtration or autoclaving. To minimize decomposition of sodium bicarbonate by decarboxylation on autoclaving, carbon dioxide is passed through the solution in its final container, which is then hermetically sealed and autoclaved. The sealed container should not be opened for at least 2 hours after it has returned to ambient temperature, to allow time for the complete reformation of the bicarbonate from the carbonate produced during the heating process. Aqueous solutions of sodium bicarbonate stored in glass containers may develop deposits of small glass particles. Sediments of calcium carbonate with traces of magnesium or other metal carbonates have been found in injections sterilized by autoclaving; these are due to impurities in the bicarbonate or to extraction of calcium and magnesium ions from the glass container. Sedimentation may be retarded by the inclusion of 0.01–0.02% disodium edetate.(37–39) Sodium bicarbonate is stable in dry air but slowly decomposes in moist air and should therefore be stored in a well-closed container in a cool, dry place.

Sodium bicarbonate reacts with acids, acidic salts, and many alkaloidal salts, with the evolution of carbon dioxide. Sodium bicarbonate can also intensify the darkening of salicylates. In powder mixtures, atmospheric moisture or water of crystallization from another ingredient is sufficient for sodium bicarbonate to react with compounds such as boric acid or alum. In liquid mixtures containing bismuth subnitrate, sodium bicarbonate reacts with the acid formed by hydrolysis of the bismuth salt. In solution, sodium bicarbonate has been reported to be incompatible with many drug substances such as ciprofloxacin,(40,41) amiodarone,(42) nicardipine,(43) and levofloxacin.(44)
Sodium bicarbonate is manufactured either by passing carbon dioxide into a cold saturated solution of sodium carbonate, or by the ammonia–soda (Solvay) process, in which first ammonia and then carbon dioxide is passed into a sodium chloride solution to precipitate sodium bicarbonate while the more soluble ammonium chloride remains in solution.
Sodium bicarbonate is used in a number of pharmaceutical formulations including injections and ophthalmic, otic, topical, and oral preparations. Sodium bicarbonate is metabolized to the sodium cation, which is eliminated from the body by renal excretion, and the bicarbonate anion, which becomes part of the body’s bicarbonate store. Any carbon dioxide formed is eliminated via the lungs. Administration of excessive amounts of sodium bicarbonate may thus disturb the body’s electrolyte balance, leading to metabolic alkalosis or possibly sodium overload with potentially serious consequences. The amount of sodium present in antacids and effervescent formulations has been sufficient to exacerbate chronic heart failure, especially in elderly patients.(45) Orally ingested sodium bicarbonate neutralizes gastric acid with the evolution of carbon dioxide and may cause stomach cramps and flatulence. When used as an excipient, sodium bicarbonate is generally regarded as an essentially nontoxic and nonirritant material. LD50 (mouse, oral): 3.36 g/kg(46) LD50 (rat, oral): 4.22 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Eye protection and gloves are recommended.
GRAS listed. Accepted for use as a food additive in Europe. Included in the FDA Inactive Ingredients Database (injections; ophthalmic preparations; oral capsules, solutions, and tablets). Included in parenteral (intravenous infusions and injections) and nonparenteral medicines (chewing gums; ear drops; eye lotions; oral capsules, chewable tablets, effervescent powders, effervescent tablets, granules, soluble tablets, orodispersible tablets, and tablets; suppositories and suspensions) licensed in the UK.
Potassium bicarbonate.
Each gram of sodium bicarbonate represents approximately 11.9 mmol of sodium and of bicarbonate. Each gram of sodium bicarbonate will neutralize 12 mEq of gastric acid in 60 minutes. The yield of carbon dioxide from sodium bicarbonate is approximately 52% by weight. Three molecules of sodium bicarbonate are required to neutralize one molecule of citric acid, and two molecules of sodium bicarbonate to neutralize one molecule of tartaric acid. A specification for sodium bicarbonate is contained in the Food Chemicals Codex (FCC).(47) The EINECS number for sodium bicarbonate is 205-633-8. The PubChem Compound ID (CID) for sodium bicarbonate includes 516892 and 24192197