

Name: Thymol
CAS No: Thymol [89-83-8]
BP: Thymol JP: Thymol PhEur: Thymol USP-NF: Thymol
Acido trimico; 3-p-cymenol; p-cymen-3-ol; Flavinol; 3-hydroxy-pcymene; 3-hydroxy-1-methyl-4-isopropylbenzene; Intrasol; isopropyl cresol; isopropyl-m-cresol; 6-isopropyl-m-cresol; isopropyl metacresol; 2-isopropyl-5-methylphenol; 1-methyl-3-hydroxy-4- isopropylbenzene; 5-methyl-2-isopropylphenol; 5-methyl-2-(1- methylethyl) phenol; Medophyll; thyme camphor; thymic acid; mthymol; thymolum; timol.
Thymol [89-83-8]
C10H14O 150.24
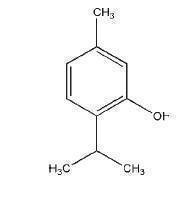
Thymol is a phenolic antiseptic, which has antibacterial and antifungal activity. However, it is not suitable for use as a preservative in pharmaceutical formulations because of its low aqueous solubility. The antimicrobial activity of thymol against eight oral bacteria has been studied in vitro. Inhibitory activity was noted against almost all organisms, and a synergistic effect was observed for combinations of thymol and eugenol, and of thymol and carvacrol.(1) The activity of thymol against bacteria commonly involved in upper respiratory tract infections has also been shown.(2) Thymol is a more powerful disinfectant than phenol, but its low water solubility, its irritancy to tissues, and its inactivation by organic material, such as proteins, limit its use as a disinfectant. Thymol is chiefly used as a deodorant in antiseptic mouthwashes, gargles, and toothpastes, such as in Compound Thymol Glycerin BP, in which it has no antiseptic action. Thymol is also a true antioxidant and has been used at concentrations of 0.01% as an antioxidant for halothane, trichloroethylene, and tetrachloroethylene. The antioxidant activity of thymol(3,4) and thymol analogues(3) has been described. More recently, thymol has been shown to enhance the in vitro percutaneous absorption of a number of drugs, including 5- fluorouracil,(5) piroxicam,(6) propranolol,(7) naproxen,(8) and tamoxifen.(9) Studies have also demonstrated that the melting point of lidocaine is significantly lowered when it is mixed with thymol.(10,11) The inhalation of thymol, in combination with other volatile substances, is used to alleviate the symptoms of colds, coughs, and associated respiratory disorders. Externally, thymol has been used in dusting powders for the treatment of fungal skin infections; thymol has been shown to have synergistic antifungal effects when combined with ketoconazole.(12) Thymol was formerly used in the treatment of hookworm infections but has now been superseded by less toxic substances. In dentistry, thymol has been mixed with phenol and camphor to prepare cavities before filling, and mixed with zinc oxide to form a protective cap for dentine. Thymol has been included in food, perfume, and cosmetic products, and has also been used as a pesticide and fungicide.
Thymol occurs as colorless or often large translucent crystals, or as a white crystalline powder with a herbal odor (aromatic and thymelike) and a pungent caustic taste.
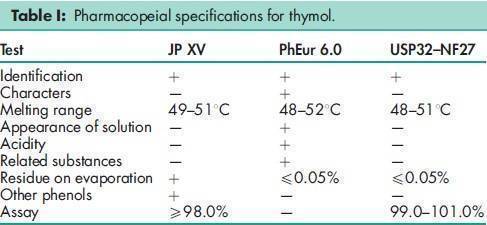
See Table I.
Acidity/alkalinity A 4% solution in ethanol (50%) is neutral to litmus; a 1% solution in water has a pH of 7. Boiling point About 2338C. Density 0.97 g/cm3 at 258C; has a greater density than water, but when liquefied by fusion is less dense than water. Dissociation constant pKa = 10.6 at 208C Melting point 48–518C, but, once melted, remains liquid at a considerably lower temperature. Partition coefficient log (octanol/water) = 3.3 Phenol coefficient About 50. Refractive index nD 25 = 0.15204; nD 20 = 0.15227. Solubility Soluble 1 in 0.7–1.0 of chloroform; 1 in 1 of ethanol (95%); 1 in 1.5 of ether, glacial acetic acid; 1 in 1.7–2.0 of olive oil; 1 in 1000 of water. Freely soluble in essential oils, fixed oils, and fats. Sparingly soluble in glycerin. Dissolves in dilute solutions of alkali hydroxides, forming salts that have increased solubility but whose solutions darken on standing. Vapor pressure 0.04mmHg at 208C Volatility Appreciable volatility at 1008C; volatile in water vapor at 258C.
Thymol should be stored in well-closed, light-resistant containers, in a cool, dry, place. Thymol is affected by light.
Thymol is incompatible with iodine, alkalis, and oxidizing agents. It liquefies, or forms soft masses, on trituration with acetanilide, antipyrine, camphor, monobromated camphor, chloral hydrate, menthol, phenol, or quinine sulfate. The antimicrobial activity of thymol is reduced in the presence of proteins.
Thymol is obtained from the volatile oil of thyme (Thymus vulgaris Linne´ (Fam. Labiatae)) by fractional distillation followed by extraction and recrystallization. Thyme oil yields about 20–30% thymol. Thymol may also be produced synthetically from pcymene, menthone, or piperitone, or by the interaction of m-cresol with isopropyl chloride.
Thymol is used in cosmetics, foods, and pharmaceutical applications as an excipient. However, thymol may be irritating when inhaled or following contact with the skin or eyes. It may also cause abdominal pain and vomiting, and sometimes stimulation followed by depression of the central nervous system following oral consumption; fats and alcohol increase absorption and aggravate symptoms. Respiratory arrest, attributed to acute nasal congestion and edema, has been reported in a 3-week-old patient due to the erroneous intranasal application of Karvol, a combination product that includes thymol. The patient recovered, but it was recommended that inhalation decongestants should not be used in children under the age of 5 years.(13) LD50 (guinea pig, oral): 0.88 g/kg(14) LD50 (mouse, IP): 0.11 g/kg LD50 (mouse, IV): 0.1 g/kg LD50 (mouse, oral): 0.64 g/kg LD50 (mouse, SC): 0.243 g/kg LD50 (rat, oral): 0.98 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Special precautions should be taken to avoid inhalation, or contact with the skin or eyes. Eye protection and gloves are recommended. When thymol is heated to decomposition, carbon dioxide and carbon monoxide are formed.
GRAS listed. Included in the FDA Inactive Ingredients Database (inhalation, liquid; oral, powder for solution). Included in nonparenteral medicines (topical creams and ointments) licensed in the UK. Included in the Canadian List of Acceptable Nonmedicinal Ingredients.
Menthol.
The EINECS number for thymol is 201-944-8. The PubChem Compound ID (CID) for thymol is 6989.