

Name: Sodium Chloride
CAS No: Sodium chloride [7647-14-5]
BP: Sodium Chloride JP: Sodium Chloride PhEur: Sodium Chloride USP: Sodium Chloride
Alberger; chlorure de sodium; common salt; hopper salt; natrii chloridum; natural halite; rock salt; saline; salt; sea salt; table salt
Sodium chloride [7647-14-5]
NaCl 58.44
See Section 4.
Sodium chloride is widely used in a variety of parenteral and nonparenteral pharmaceutical formulations, where the primary use is to produce isotonic solutions. Sodium chloride has been used as a lubricant and diluent in capsules and direct-compression tablet formulations in the past,(1–5) although this practice is no longer common. Sodium chloride has also been used as a channeling agent(6,7) and as an osmotic agent(8,9) in the cores of controlled-release tablets. It has been used as a porosity modifier in tablet coatings,(10) and to control drug release from microcapsules.(11,12) The addition of sodium chloride to aqueous spray-coating solutions containing hydroxypropyl cellulose or hypromellose suppresses the agglomeration of crystalline cellulose particles.(13) Sodium chloride can also be used to modify drug release from gels(14) and from emulsions.(15) It can be used to control micelle size,(16–18) and to adjust the viscosity of polymer dispersions by altering the ionic character of a formulation.(19,20)

Sodium chloride occurs as a white crystalline powder or colorless crystals; it has a saline taste. The crystal lattice is a face-centered cubic structure. Solid sodium chloride contains no water of crystallization although, below 08C, salt may crystallize as a dihydrate.
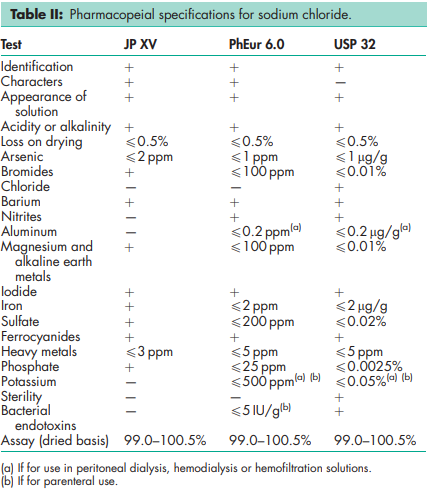
See Table II.
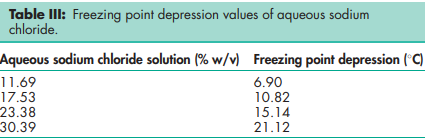
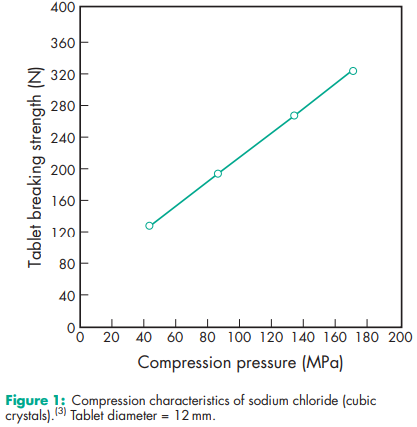
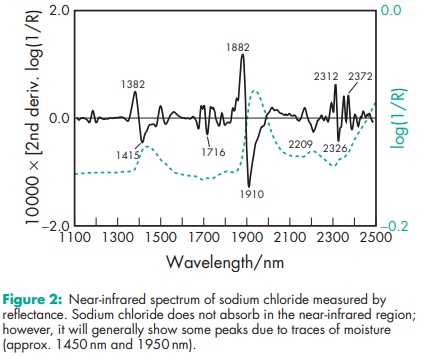
Acidity/alkalinity pH = 6.7–7.3 (saturated aqueous solution) Angle of repose 388 for cubic crystals Boiling point 14138C Compressibility With sodium chloride powder of less than 30 mm particle size, tablets are formed by plastic deformation; above this size, both plastic deformation and fracture occur.(1,3,4) See also Figure 1. Density 2.17 g/cm3 ; 1.20 g/cm3 for saturated aqueous solution. Density (bulk) 0.93 g/cm3 Density (tapped) 1.09 g/cm3 Dielectric constant 5.9 at 1 MHz Freezing point depression Hardness (Mohs) 2–2.5 Hygroscopicity Hygroscopic above 75% relative humidity. Melting point 8048C NIR spectra see Figure 2. Osmolarity A 0.9% w/v aqueous solution is iso-osmotic with serum. Refractive index nD 20 = 1.343 for a 1 M aqueous solution. Solubility see Table IV. Thermal conductivity 1.15 Wm/K at 273 K Specific heat capacity 854 J/kg/K Vapor pressure 133.3 Pa at 8658C for solid; 1759.6 Pa at 208C for a saturated aqueous solution (equivalent to 75.3% relative humidity). Viscosity A 10% w/v solution has a viscosity of 1.19 mPa s (1.19 cP).


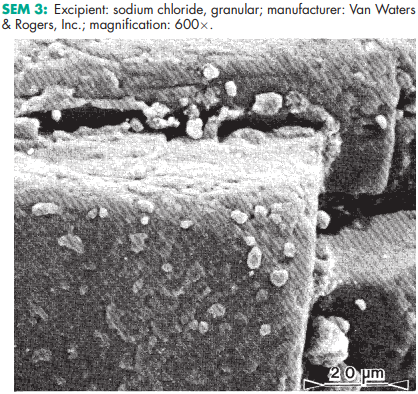

Aqueous sodium chloride solutions are stable but may cause the separation of glass particles from certain types of glass containers. Aqueous solutions may be sterilized by autoclaving or filtration. The solid material is stable and should be stored in a well-closed container, in a cool, dry place. It has been shown that the compaction characteristics and the mechanical properties of tablets are influenced by the relative humidity of the storage conditions under which sodium chloride was kept.(21,22
Aqueous sodium chloride solutions are corrosive to iron. They also react to form precipitates with silver, lead, and mercury salts. Strong oxidizing agents liberate chlorine from acidified solutions of sodium chloride. The solubility of the antimicrobial preservative methylparaben is decreased in aqueous sodium chloride solutions(23) and the viscosity of carbomer gels and solutions of hydroxyethyl cellulose or hydroxypropyl cellulose is reduced by the addition of sodium chloride
Sodium chloride occurs naturally as the mineral halite. Commercially, it is obtained by the solar evaporation of sea water, by mining, or by the evaporation of brine from underground salt deposit
Sodium chloride is the most important salt in the body for maintaining the osmotic tension of blood and tissues. About 5–12 g of sodium chloride is consumed daily, in the normal adult diet, and a corresponding amount is excreted in the urine. As an excipient, sodium chloride may be regarded as an essentially nontoxic and nonirritant material. However, toxic effects following the oral ingestion of 0.5–1.0 g/kg body-weight in adults may occur. The oral ingestion of larger quantities of sodium chloride, e.g. 1000 g in 600 mL of water,(24) is harmful and can induce irritation of the gastrointestinal tract, vomiting, hypernatremia, respiratory distress, convulsions, or death. In rats, the minimum lethal intravenous dose is 2.5 g/kg bodyweight. LD50 (mouse, IP): 6.61 g/kg(25) LD50 (mouse, IV): 0.65 g/kg LD50 (mouse, oral): 4.0 g/kg LD50 (mouse, SC): 3.0 g/kg LD50 (rat, oral): 3.0 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. If heated to high temperatures, sodium chloride evolves a vapor irritating to the eyes.
GRAS listed. Included in the FDA Inactive Ingredients Database (injections; inhalations; nasal, ophthalmic, oral, otic, rectal, and topical preparations). Included in nonparenteral and parenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients
Potassium chloride
Sodium chloride is one of the materials that have been selected for harmonization by the Pharmacopeial Discussion Group. For further information see the General Information Chapter <1196> in the USP32–NF27, the General Chapter 5.8 in PhEur 6.0, along with the ‘State of Work’ document on the PhEur EDQM website, and also the General Information Chapter 8 in the JP XV. Domestic table salt may contain sodium iodide (as a prophylactic substance against goiter) and agents such as magnesium carbonate, calcium phosphate, or starch, which reduce the hygroscopic characteristics of the salt and maintain the powder in a free-flowing state. Food-grade dendritic salt, which is porous, can be used as an absorbent for liquid medications, and as a tablet diluent in specific formulations. Each gram of sodium chloride represents approximately 17.1 mmol of sodium and 17.1 mmol of chloride; 2.54 g of sodium chloride is approximately equivalent to 1 g of sodium. A saturated solution of sodium chloride can be used as a constant-humidity solution; at 258C, a relative humidity of 75% is produced. A specification for sodium chloride is contained in the Food Chemicals Codex (FCC).(26) The EINECS number for sodium chloride is 231-598-3. The PubChem Compound ID (CID) for sodium chloride is 5234.