

Name: Sodium Hyaluronate
CAS No: Sodium hyaluronate [9067-32-7]
BP: Sodium Hyaluronate PhEur: Sodium Hyaluronate
Hyaluronan; hyaluronate sodium; natrii hyaluronas; RITA HA C1-C.
Sodium hyaluronate [9067-32-7]
(C14H20NO11Na)n (401.3)n
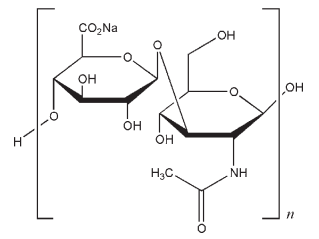
Sodium hyaluronate is the predominant form of hyaluronic acid at physiological pH. The name hyaluronan is used when the polysaccharide is mentioned in general terms, and in the literature the terms hyaluronic acid and sodium hyaluronate are used interchangeably. Hyaluronan is used therapeutically to treat osteoarthritis in the knee, and is an effective treatment for arthritic pain.(1) Crosslinked hyaluronan gels are used as drug delivery systems.(2) Hyaluronan is the most common negatively charged glycosaminoglycan in the human vitreous humor, and is known to interact with polymeric and liposomal DNA complexes,(3) where hyaluronan solutions have been shown to decrease the cellular uptake of complexes.(4) This is useful for enhancing the availability and retention time of drugs administered to the eye. It is immunoneutral, which makes it useful for the attachment of biomaterials for use in tissue engineering and drug delivery systems;(5) it also has important applications in the fields of vascosurgery and vascosupplementation.(6)
The PhEur 6.3 describes sodium hyaluronate as the sodium salt of hyaluronic acid, a glycosaminoglycan consisting of D-glucuronic acid and N-acetyl-D-glucosamine disaccharide units. Sodium hyaluronate occurs as white to off-white powder or granules. It is very hygroscopic.
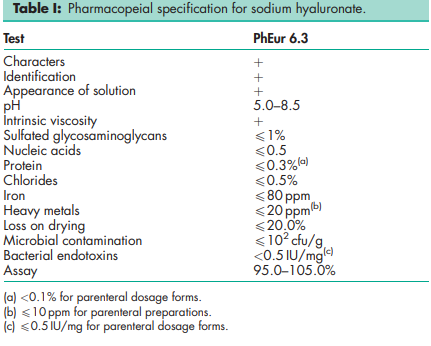
See Table 1.
Acidity/alkalinity pH = 5.0–8.5 (0.5% w/v aqueous solution) Solubility Soluble in water, although speed of dissolution depends upon molecular weight (higher molecular weights are slower to dissolve, although this process can be increased by gentle agitation). Slightly soluble in mixtures of organic solvents with water.(7)
Sodium hyaluronate should be stored in a cool, dry place in tightly sealed containers. The powder is stable for 3 years if stored in unopened containers.
Sodium hyaluronate occurs naturally in vitreous humor, serum, chicken combs, shark skin, and whale cartilage; it is usually extracted and purified from chicken combs. It may also be manufactured by fermentation of selected Streptococcus zooepidemicus bacterial strains; sodium hyaluronate is removed from the fermentation medium by filtration and purified by ultrafiltration. It is then precipitated with an organic solvent and dried.
Sodium hyaluronate is used in cosmetics and in topical, parenteral, and ophthalmic pharmaceutical formulations. It is generally regarded as a relatively nontoxic and nonirritant material. Sodium hyaluronate has been reported to be an experimental teratogen.(8) LD50 (mouse, IP): 1.5 g/kg(8) LD50 (rabbit, IP): 1.82 g/kg LD50 (rat, IP): 1.77 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. When heated to decomposition, sodium hyaluronate emits toxic fumes of Na2O.
Included in the FDA Inactive Ingredients Database (topical gel preparation).
Hyaluronic acid. Hyaluronic acid Molecular weight Hyaluronic acid molecules have a molecular weight of 300–2000 kDa as the number of repeating disaccharide units in each molecule is variable. In its natural form, hyaluronic acid exists as a high-molecular-weight polymer of 106 –107 Da. CAS number [9067-32-7] Appearance Hyaluronic acid appears as a white to off-white powder or granules. Comments Hyaluronic acid is used as an adjuvant for ophthalmic drug delivery,(9) and has been found to enhance the absorption of drugs and proteins via mucosal tissue.(10) It has also been used experimentally in controlled-release films that are suitable for application to surgical sites for the prevention of adhesion formation,(11) and in matrix formulations used in gene delivery systems.(12) The EINECS number for hyaluronic acid is 232-678- 0.
Microspheres prepared from hyaluronan esters have been evaluated for the vaginal administration of calcitonin in the treatment of postmenopausal osteoporosis.(13) Microspheres prepared from hyaluronan esters have also been used experimentally as delivery devices for nerve growth factors,(14) and as a nasal delivery system for insulin.(15) An N-(2-hydroxypropyl)methacrylamide (HPMA)–hyaluronan polymeric drug delivery system has been used for the targeted delivery of doxorubicin to cancer cells. This copolymer exhibited increased toxicity due to hyaluronan receptor-mediated uptake of the macromolecular drug.(16) The EINECS number for sodium hyaluronate is232-678-0. The PubChem Compound ID (CID) for sodium hyaluronate is 3084049