

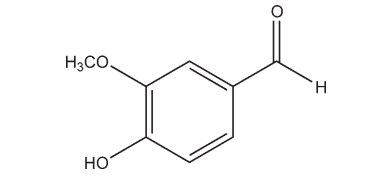
Name: Vanillin
CAS No: 4-Hydroxy-3-methoxybenzaldehyde [121-33-5]
BP: Vanillin PhEur: Vanillin USP-NF: Vanillin
4-Hydroxy-m-anisaldehyde; p-hydroxy-m-methoxybenzaldehyde; 3-methoxy-4-hydroxybenzaldehyde; methylprotocatechuic aldehyde; Rhovanil; vanillic aldehyde; vanillinum.
4-Hydroxy-3-methoxybenzaldehyde [121-33-5]
C8H8O3 ,152.15
Vanillin is widely used as a flavor in pharmaceuticals, foods, beverages, and confectionery products, to which it imparts a characteristic taste and odor of natural vanilla. It is also used in perfumes, as an analytical reagent and as an intermediate in the synthesis of a number of pharmaceuticals, particularly methyldopa. Additionally, it has been investigated as a potential therapeutic agent in sickle cell anemia(1) and is claimed to have some antifungal properties.(2) In food applications, vanillin has been investigated as a preservative.(3,4) As a pharmaceutical excipient, vanillin is used in tablets, solutions (0.01–0.02% w/v), syrups, and powders to mask the unpleasant taste and odor characteristics of certain formulations, such as caffeine tablets and polythiazide tablets. It is similarly used in film coatings to mask the taste and odor of vitamin tablets. Vanillin has also been investigated as a photostabilizer in furosemide 1% w/v injection, haloperidol 0.5% w/v injection, and thiothixene 0.2% w/v injection.(5)
White or cream, crystalline needles or powder with characteristic vanilla odor and sweet taste.
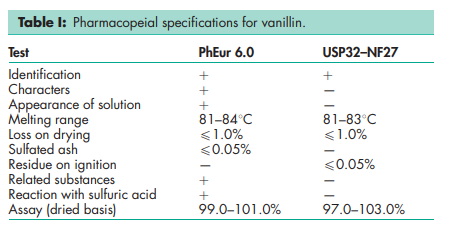
See Table I
Acidity/alkalinity Aqueous solutions are acid to litmus. Boiling point 284–2858C (with decomposition) Density (bulk) 0.6 g/cm3 Flash point 1538C (closed cup) Melting point 81–838C NIR spectra see Figure 1. Solubility see Table II. Specific gravity 1.056 (liquid)
Vanillin oxidizes slowly in moist air and is affected by light. Solutions of vanillin in ethanol decompose rapidly in light to give a yellow-colored, slightly bitter tasting solution of 6,60 -dihydroxy5,50 -dimethoxy-1,10 -biphenyl-3,30 -dicarbaldehyde. Alkaline solutions also decompose rapidly to give a brown-colored solution. However, solutions stable for several months may be produced by adding sodium metabisulfite 0.2% w/v as an antioxidant.(6) The bulk material should be stored in a well-closed container, protected from light, in a cool, dry place.
Incompatible with acetone, forming a brightly colored compound.(7) A compound practically insoluble in ethanol is formed with glycerin
Vanillin occurs naturally in many essential oils and particularly in the pods of Vanilla planifolia and Vanilla tahitensis. Industrially, vanillin is prepared from lignin, which is obtained from the sulfite wastes produced during paper manufacture. Lignin is treated with alkali at elevated temperature and pressure, in the presence of a catalyst, to form a complex mixture of products from which vanillin is isolated. Vanillin is then purified by successive recrystallizations. Vanillin may also be prepared synthetically by condensation, in weak alkali, of a slight excess of guaiacol with glyoxylic acid at room temperature. The resultant alkaline solution, containing 4- hydroxy-3-methoxymandelic acid is oxidized in air, in the presence of a catalyst, and vanillin is obtained by acidification and simultaneous decarboxylation. Vanillin is then purified by successive recrystallizations.
There have been few reports of adverse reactions to vanillin, although it has been speculated that cross-sensitization with other structurally similar molecules, such as benzoic acid, may occur.(8) Adverse reactions that have been reported include contact dermatitis(9) and bronchospasm caused by hypersensitivity.(10) The WHO has allocated an estimated acceptable daily intake for vanillin of up to 10 mg/kg body-weight.(11) LD50 (guinea pig, IP): 1.19 g/kg(12) LD50 (guinea pig, oral): 1.4 g/kg LD50 (mouse, IP): 0.48 g/kg LD50 (rat, IP): 1.16 g/kg LD50 (rat, oral): 1.58 g/kg LD50 (rat, SC): 1.5 g/kg
Observe normal precautions appropriate to the quantity of material handled. Eye protection is recommended. Heavy airborne concentrations of dust may present an explosion hazard.
GRAS listed. Included in the FDA Inactive Ingredients Database (oral solutions, suspensions, syrups, and tablets). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Ethyl vanillin.
One part of synthetic vanillin is equivalent to 400 parts of vanilla pods. The EINECS number for vanillin is 204-465-2. The PubChem Compound ID (CID) for vanillin is 1183.