

Name: Sodium Lactate
CAS No: Sodium lactate [72-17-3]
BP: Sodium Lactate Solution PhEur: Sodium Lactate Solution USP: Sodium Lactate Solution
E325; 2-hydroxypropanoic acid monosodium salt; Lacolin; lactic acid monosodium salt; lactic acid sodium salt; natrii lactatis solutio; Patlac; Purasal; Ritalac NAL; sodium a-hydroxypropionate.
Sodium lactate [72-17-3]
C3H5NaO3 112.06
The PhEur 6.0 and USP 32 describe sodium lactate solution as a mixture of the enantiomers of sodium 2-hydroxypropanoate in approximately equal proportions.
Sodium lactate is widely used in cosmetics,(1,2) food products and pharmaceutical applications including parenteral and topical formulations. Therapeutically, sodium lactate is used in infusions as a component of Ringer-lactate solution; as an alternative for sodium hydrogencarbonate in light acidosis; as a rehydrating agent; and as a carrier for electrolyte concentrates or medicines in perfusion/ infusion solutions
Sodium lactate occurs as a clear, colorless, slightly syrupy liquid. It is odorless, or has a slight odor with a characteristic saline taste. It is hygroscopic.
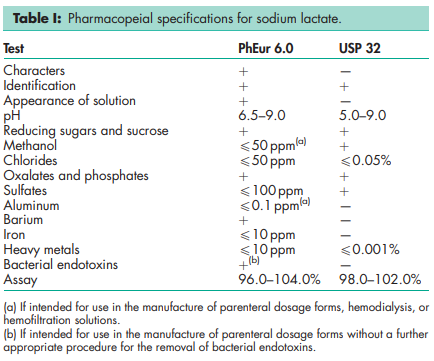
See Table I.
Acidity/alkalinity pH = 7 for an aqueous solution. Boiling point 1128C Hygroscopicity Very hygroscopic. Melting point 178C with decomposition at 1408C. Solubility Miscible with ethanol (95%), and with water. Specific gravity 1.311.34
Sodium lactate should be stored in a well-closed container in a cool, dry, place. Sodium lactate is combustible and decomposes upon heating.
See Lactic Acid.
See Lactic Acid.
Sodium lactate occurs naturally in the body and is involved in physiological processes. It is generally regarded as a relatively nontoxic and nonirritant material when used as an excipient. Low concentrations are well tolerated by skin and eye mucosa, although higher concentrations should be avoided. LD50 (rat, IP): 2 g/kg(3)
Observe normal precautions appropriate to the circumstances and quantity of material handled. Sodium lactate may cause eye irritation. When heated to decomposition, sodium lactate emits toxic fumes of Na2O.(3)
GRAS listed (not for infant formulas). Included in the FDA Inactive Ingredient Database (epidural, IM, IV, and SC injections; oral suspensions; topical gels and solutions). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Lactic Acid.
Generally, the commercially available product is a mixture with water containing 70–80% sodium lactate. The EINECS number for sodium lactate is 200-772-0. The PubChem Compound ID (CID) for sodium lactate is 23666456.