

Name: Butylated Hydroxyanisole
CAS No: 2-tert-Butyl-4-methoxyphenol [25013-16-5]
BP: Butylated Hydroxyanisole PhEur: Butylhydroxyanisole USP-NF: Butylated Hydroxyanisole
BHA; tert-butyl-4-methoxyphenol; butylhydroxyanisolum; 1,1- dimethylethyl-4-methoxyphenol; E320; Nipanox BHA; Nipantiox 1-F; Tenox BHA
2-tert-Butyl-4-methoxyphenol [25013-16-5]
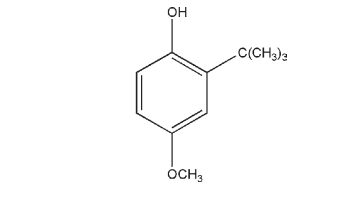
C11H16O2 180.25 The PhEur 6.0 describes butylated hydroxyanisole as 2-(1,1- dimethylethyl)-4-methoxyphenol containing not more than 10% of 3-(1,1-dimethylethyl)-4-methoxyphenol.
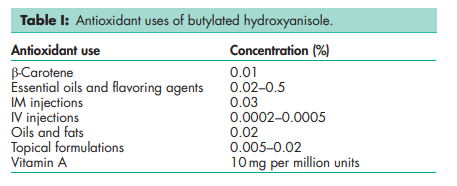
Butylated hydroxyanisole is an antioxidant (see Table I) with some antimicrobial properties.(1–3) It is used in a wide range of cosmetics, foods, and pharmaceuticals. When used in foods, it is used to delay or prevent oxidative rancidity of fats and oils and to prevent loss of activity of oil-soluble vitamins. Butylated hydroxyanisole is frequently used in combination with other antioxidants, particularly butylated hydroxytoluene and alkyl gallates, and with sequestrants or synergists such as citric acid. FDA regulations direct that the total content of antioxidant in vegetable oils and direct food additives shall not exceed 0.02% w/w (200 ppm) of fat or oil content or essential (volatile) oil content of food. USDA regulations require that the total content of antioxidant shall not exceed 0.01% w/w (100 ppm) of any one antioxidant or 0.02% w/w combined total of any antioxidant combination in animal fats. Japanese regulations allow up to 1 g/kg in animal fats.
Butylated hydroxyanisole occurs as a white or almost white crystalline powder or a yellowish-white waxy solid with a faint, characteristic aromatic odor
See Table II.

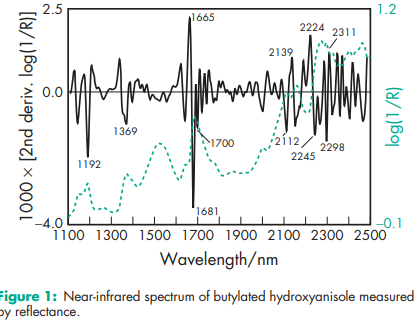
Antimicrobial activity Activity is similar to that of the phydroxybenzoate esters (parabens). The greatest activity is against molds and Gram-positive bacteria, with less activity against Gram-negative bacteria. Boiling point 2648C at 745 mmHg Density (true) 1.117 g/cm3 Flash point 1308C Melting point 478C (for pure 2-tert-butyl-4-methoxyphenol); see also Section 18. NIR spectra see Figure 1. Solubility Practically insoluble in water; soluble in methanol; freely soluble in 550% aqueous ethanol, propylene glycol, chloroform, ether, hexane, cottonseed oil, peanut oil, soybean oil, glyceryl monooleate, and lard, and in solutions of alkali hydroxides. Viscosity (kinematic) 3.3 mm2 /s (3.3 cSt) at 998C.
Exposure to light causes discoloration and loss of activity. Butylated hydroxyanisole should be stored in a well-closed container, protected from light, in a cool, dry place.
Butylated hydroxyanisole is phenolic and undergoes reactions characteristic of phenols. It is incompatible with oxidizing agent and ferric salts. Trace quantities of metals and exposure to light cause discoloration and loss of activity.
Prepared by the reaction of p-methoxyphenol with isobutene.
Butylated hydroxyanisole is absorbed from the gastrointestinal tract and is metabolized and excreted in the urine with less than 1% unchanged within 24 hours of ingestion.(4) Although there have been some isolated reports of adverse skin reactions to butylated hydroxyanisole,(5,6) it is generally regarded as nonirritant and nonsensitizing at the levels employed as an antioxidant. Concern over the use of butylated hydroxyanisole has occurred following long-term animal feeding studies. Although previous studies in rats and mice fed butylated hydroxyanisole at several hundred times the US-permitted level in the human diet showed no adverse effects, a study in which rats, hamsters, and mice were fed butylated hydroxyanisole at 1–2% of the diet produced benign and malignant tumors of the forestomach, but in no other sites. However, humans do not have any region of the stomach comparable to the rodent forestomach and studies in animals that also do not have a comparable organ (dogs, monkeys, and guinea pigs) showed no adverse effects. Thus, the weight of evidence does not support any relevance to the human diet where butylated hydroxyanisole is ingested at much lower levels.(7) The WHO acceptable daily intake of butylated hydroxyanisole has been set at 500 mg/kg body-weight.(7) LD50 (mouse, oral): 1.1–2.0 g/kg(8) LD50 (rabbit, oral): 2.1 g/kg LD50 (rat, IP): 0.88 g/kg LD50 (rat, oral): 2.0 g/kg
Observe normal precautions appropriate to the circumstances and quantity of material handled. Butylated hydroxyanisole may be irritant to the eyes and skin and on inhalation. It should be handled in a well-ventilated environment; gloves and eye protection are recommended. On combustion, toxic fumes may be given off.
GRAS listed. Accepted as a food additive in Europe. Included in the FDA Inactive Ingredients Database (IM and IV injections, nasal sprays, oral capsules and tablets, and sublingual, rectal, topical, and vaginal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Nonmedicinal Ingredients.
Butylated hydroxytoluene.
The commercially available material can have a wide melting point range (47–578C) owing to the presence of varying amounts of 3- tert-butyl-4-methoxyphenol. Tenox brands contain 0.1% w/w citric acid as a stabilizer. A specification for butylated hydroxyanisole is contained in the Food Chemicals Codex (FCC).(9) The EINECS number for butylated hydroxyanisole is 246-563- 8. The PubChem Compound ID (CID) for butylated hydroxyanisole includes 8456 and 11954184.